Acropustulosis of
Infancy
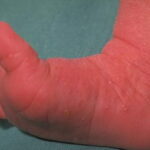
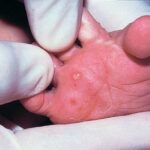
Infantile acropustulosis is a recurrent, self-limited, pruritic, vesicopustular eruption of the palms and the soles occurring in young children during the first 2-3 years of life. Newly described in 1979, infantile acropustulosis is probably much more common than the scarcity of reports would imply
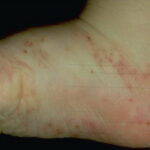
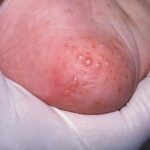
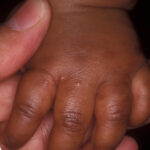
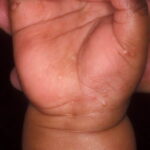
- The hands and the feet are always involved in infantile acropustulosis, usually on the palms, the soles, and the lateral surfaces. Lesions may occur on the dorsal aspects of the hands and the feet as well as the trunk, the scalp, and the face.
- Infantile acropustulosis lesions begin as small macules or papules that then form distinct, noncoalescing vesicles and pustules
- They heal with macular hyperpigmentation.
- No other organ systems are involved in infantile acropustulosis,
-
Causes
The cause of infantile acropustulosis is unknown. Scabies as a preceding or concomitant infestation is well documented in some cases. Many children are undoubtedly misdiagnosed as having scabies and treated with lindane or permethrin without any confirmatory scrapings. No other infectious agent has been documented
Treatment
-
-
Medical Care
Treatment is often unnecessary because of the self-limited nature of infantile acropustulosis.
- Topical steroids and oral dapsone have been used successfully, if justified in more difficult cases.
- Topical pramoxine preparations are available without prescription for the treatment of pruritus.
- Oral antihistamines may be useful in infantile acropustulosis.
Consultations
-
In infantile acropustulosis, consult a dermatologist or a pediatric dermatologist.
Activity
-
Medication
-
-
and 2) have been used successfully for
-
control of pruritus. Children who are
-
extremely symptomatic may be treated
-
with dapsone.
Topical steroids
-
These agents provide symptomatic relief of pruritus.
Betamethasone (Diprolene, Betatrex)
-
For inflammatory dermatoses responsive to
-
steroids. Decreases inflammation by
-
suppressing migration of
-
polymorphonuclear leukocytes and
-
reversing capillary permeability. Use
-
fluorinated topical steroids with caution in children.
Adult
-
Pediatric
Apply thin film to affected areas bid; occlusion increases effectiveness; avoid wraps that may present choking hazard
None reported
Documented hypersensitivity; paronychia; cellulitis; impetigo; angular cheilitis; erythrasma; erysipelas; rosacea; perioral dermatitis; acne
Pregnancy
-
C - Fetal risk revealed in studies in animals but not established or not studied in humans; may use if benefits outweigh risk to fetus
Precautions
Application over large surface areas may cause systemic absorption and adrenal suppression; do not use on skin with decreased circulation; can cause atrophy of groin, face, and axillae; if infection develops and is not responsive to antibiotic treatment, discontinue until infection is under control
Antibiotics
Diaminodiphenylsulfone antibiotics have been used as anti-inflammatory agents.
Dapsone (Avlosulfon)
Bactericidal and bacteriostatic against mycobacteria; mechanism of action is similar to that of sulfonamides where competitive antagonists of PABA prevent formation of folic acid, inhibiting bacterial growth. Used mainly to treat leprosy and dermatitis herpetiformis. Has antineutrophil and anti-inflammatory properties.
Adult
Pediatric
1-2 mg/kg/d PO; not to exceed 100 mg
May inhibit anti-inflammatory effects of clofazimine; hematologic reactions may increase with folic acid antagonists, eg, pyrimethamine (monitor for agranulocytosis during second and third months of therapy); probenecid increases toxicity; trimethoprim with dapsone may increase toxicity of both drugs; because of increased renal clearance, levels may significantly decrease when administered concurrently with rifampin
Concomitant administration of zidovudine may increase risk of hematologic toxicity; amprenavir and saquinavir may inhibit cytochrome P4503A (CYP3A), the hepatic isoenzyme group with major activity related to dapsone metabolism, thereby leading to increased dapsone serum concentrations and potential toxicityDocumented hypersensitivity; known G-6-PD deficiency (assay for G-6-PD activity prior to initiation of therapy)
Pregnancy
C - Fetal risk revealed in studies in animals but not established or not studied in humans; may use if benefits outweigh risk to fetus
Precautions
Associated with a variety of systemic toxicities, including agranulocytosis, anemia, methemoglobinemia, hepatitis, and neuropathy; patients may experience headache and/or GI distress on initiation of therapy; perform weekly blood counts (first mo), then monthly WBC counts (6 mo), then semiannual WBC counts; discontinue if a significant reduction in platelets, leukocytes, or hematopoiesis occurs; caution in methemoglobin reductase deficiency, G-6-PD deficiency, or hemoglobin M because of high risk for hemolysis and Heinz body formation; caution in patients exposed to other agents or conditions (eg, infection, diabetic ketosis) capable of producing hemolysis; peripheral neuropathy can occur (rare); phototoxicity may occur when exposed to UV light; pancreatitis may occur; various forms of renal complications including acute renal failure, acute tubular necrosis, and oliguria have occurred with dapsone use
Antipruritics
These agents may relieve associated itching.
Pramoxine (Tronothane, Prax)
Blocks nerve conduction and impulses by inhibiting depolarization of neurons. Use 1% lotion or cream.
Adult
Pediatric
Apply to affected area prn; not to exceed 200 mg
None reported
Documented hypersensitivity; do not apply over large areas; avoid contact with eyes and nose
Pregnancy
C - Fetal risk revealed in studies in animals but not established or not studied in humans; may use if benefits outweigh risk to fetus
Precautions
Caution in patients with trauma in area to be treated
Frequency
United States
The exact incidence of infantile acropustulosis is unknown.
International
The exact incidence of infantile acropustulosis is unknown. One study from Israel reported 25 cases of infantile acropustulosis in a 9-year period, suggesting that this is not as uncommon as once thought.
Mortality/Morbidity
All cases of infantile acropustulosis spontaneously resolve in a few months to 3 years.
Race
Early reports suggested a predominance of African Americans for infantile acropustulosis. Now, acropustulosis of infancy is believed to affect all races equally.
Sex
Early reports suggested a male predominance for infantile acropustulosis. Larger series have since shown an equal distribution between males and females.
Age
Although children as old as 9 years have been reported to have infantile acropustulosis, acropustulosis of infancy typically begins between the first 2-12 months of life. Resolution by age 3 years is the norm.
Clinical
History
- The classic history of infantile acropustulosis is an infant aged 2-12 months developing pruritic erythematous macules or papules that progress into vesicles and then pustules.
- Children are fretful, irritable, and obviously uncomfortable, but otherwise healthy.
- Individual bouts of infantile acropustulosis last 7-15 days and recur in 2- to 4-week intervals.
- Often, children have been empirically treated with antiscabies medicines prior to presentation .
- The intensity and the duration of infantile acropustulosis attacks diminish with each recurrence.