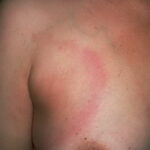
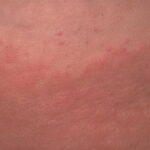
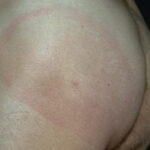
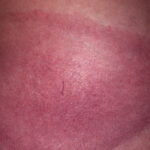
Erythema chronicum migrans
Lyme disease is a systemic infection caused by the spirochete Borrelia burgdorferi.1 The bacteria are inoculated into the skin by a tick bite, nearly always from hard-bodied ticks of the genus Ixodes.
The original descriptions of the dermatologic manifestations of Lyme disease date back to 1883 in Europe, when a German physician, Alfred Buchwald, described what is now termed acrodermatitis chronica atrophicans (ACA). Several decades later in 1912, a Swedish dermatologist, Arvid Afzelius, described erythema chronicum migrans (ECM), which currently is referred to simply as erythema migrans (EM).
It was not until 1975, when a statistically improbable cluster of childhood arthritis occurred in and around the town of Lyme, Connecticut, that the full spectrum of the disease began to be elucidated. This outbreak stimulated intense clinical and epidemiologic research that led to the discovery of the causative agent and its ecology and an expanding geographic range and list of clinical manifestations. In addition, the initial antibiotic responsiveness of the cutaneous manifestations that had been described in the 1950s in the European literature was confirmed, and the findings were extended.
In 1998, the US Food and Drug Administration (FDA) approved a protective vaccine, but the manufacturer pulled it from the market a few years later.
Pathophysiology
The pathophysiology of Lyme disease is incompletely understood. Many of its manifestations are caused by active infection by the spirochete; others may be driven by immunopathogenetic mechanisms. While any part of the body can be affected, the organism shows a distinct tropism for the skin, heart, central nervous system (CNS), joints, and eyes.
The bacteria are introduced into the skin by a bite from an infected Ixodes tick. In the northeastern and upper midwestern United States, Ixodes scapularis (sometimes termed Ixodes dammini) is the vector. In the northwestern United States, Ixodes pacificus is the vector. In other parts of the world, other Ixodes ticks serve this function. Other tick species (eg, Amblyomma americanum) and insects can carry B burgdorferi, but the vast majority of cases are believed to be caused by bites by Ixodes ticks. Note that in the southern and midcentral United States, a Lyme-like disease has been reported for which the vector appears to be A americanum.
Patients with erythema migrans from Missouri and those from New York have different clinical and microbiological aspects of the disease. B burgdorferi has not been isolated from the southern patients, although a closely related spirochete is suspected to be involved. Although some cases from the southern United States are documented with this new spirochete, called Borrelia lonestari, no organism can be isolated in the vast majority of cases of erythema migrans in this geographical area.2,3
Once in the skin, the spirochete can be overwhelmed and eliminated by host defense mechanisms, can remain viable but localized at the site of inoculation, or may disseminate via blood and lymphatics. Hematogenous dissemination can occur within days or weeks of the initial infection. The organism can travel to other parts of the skin, the heart, joints, the CNS, and other parts of the body.
Early studies showed that in roughly 10% of patients with isolated erythema migrans and no systemic symptoms, B burgdorferi or its DNA, can be detected in the bloodstream. In addition, early in the course of disease and while erythema migrans still is present, spirochetal DNA has been detected in cerebrospinal fluid, indicating early CNS penetration. This can occur even in the absence of neurologic symptoms.
Importantly, one 2005 study found that if large-volume cultures (9 mL of plasma) were performed in early presenting patients with erythema migrans, 93 (43.7%) of 213 had spirochetemia. Some of these patients had only isolated erythema migrans and no systemic symptoms.4
The organism also can persist in the skin for very long periods of time. Experimentally, the spirochete can penetrate human fibroblasts and live intracellularly, even when the extracellular medium contains ceftriaxone at concentrations well above bacteriocidal levels. While intracellular organisms have never been demonstrated in vivo, this may be one mechanism by which the organism eludes host defense mechanisms. Clinically, B burgdorferi has been cultured from skin lesions of patients with acrodermatitis chronica atrophicans 10 years after initial infection.
Because people who engage in activities that put them in risk for tick bites tend to continue those activities, reinfection is not uncommon. Patients who have had prior erythema migrans can be reinfected (meaning that the first infection has been successfully treated and they have a new infection with B burgdorferi). This has been clearly demonstrated in reinfected patients who were culture positive and had different serotypes isolated in the first and second infections. Reinfection shows similar manifestations as first infections, although a tendency towards less hematogenous spread is noted. In contrast, relapse (as opposed to reinfection) is very unusual in patients who have been treated with appropriate antimicrobials.5
Similar to syphilis, Lyme disease classically has been divided into stages; however, in individual patients, no rigid cutoffs exist between stages. Note the following:
- Early localized Lyme disease refers to isolated erythema migrans and patients who present with an undifferentiated febrile illness.
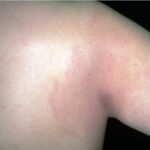
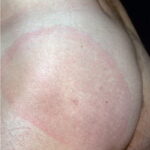
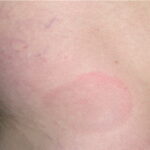
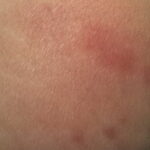
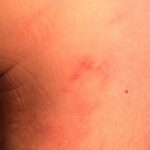
- Early disseminated disease refers to the secondary (usually hematogenously spread) skin lesions and to the extracutaneous manifestations that occur during the initial weeks to months of infection. The more common of these include lymphocytic meningitis, cranial neuropathy (usually of the seventh nerve), radiculoneuritis (more common in Europe), and carditis (often with fluctuating degrees of arteriovenous block). Borrelial lymphocytoma (BL) usually occurs during this stage (see the image below).
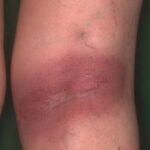
- Late Lyme disease refers to manifestations, primarily rheumatologic and neurologic, that occur months to years after initial infection. Acrodermatitis chronica atrophicans develops during this phase.
Note that while many patients present with erythema migrans, others first present with extracutaneous symptoms, either because erythema migrans never occurred or because it was not recognized by the patient or correctly diagnosed by the physician.
The last important phenomenon to appreciate is co-infection by other organisms transmitted by the same tick bite. Co-infection by ehrlichial species and Babesia microti are reported with increased frequency; in some studies, in as many as 10-15% of patients with Lyme disease. When Lyme disease is strongly suggested but some of the manifestations are atypical, these other tick-borne infections must be considered.
History
Epidemiologic context is extremely important. Since only approximately 25-30% of patients with early Lyme disease recall the tick bite, the history must be directed by the clinician towards the possibility of a tick bite. Determining where the patient lives, works, and vacations is important, as is asking about specific activities in which the patient participates at those locales. The likelihood of Lyme disease increases as the probability of a tick bite increases in a geographically endemic area.
The season is important, especially in patients with early disease. Most cases of erythema migrans occurs from May through September because the nymphal stage of the tick is responsible for most cases. For patients presenting with later cutaneous manifestations, especially acrodermatitis chronica atrophicans, questions must be directed at assessing the risk of tick bite (or prior manifestations of Lyme disease) from many years in the past.
Systemic manifestations are as follows:
- Some patients with erythema migrans have low-grade fever. A high fever, especially if accompanied by rigors or a toxic appearance, suggests coinfection with other tick-borne pathogens such as ehrlichial species or B microti, or it suggests an alternative diagnosis.
- Patients with early Lyme disease can present with a flulike illness (nonspecific febrile illness), although a paucity of respiratory and gastrointestinal tract symptoms may be present. Another diagnostic clue is prompt resolution using antiborrelial therapy.
- Fatigue, headache, myalgias, and arthralgias also may be seen in patients presenting early. Frank arthritis with objective joint swelling, redness, and pain is usually a later manifestation but can occur in the early disseminated phase.
- All systemic symptoms may occur in patients with erythema migrans.
- In patients with later cutaneous manifestations of Lyme disease, borrelial lymphocytoma, and acrodermatitis chronica atrophicans, other manifestations of the disease may coexist or may have occurred in the past. Thus, a history of Bell palsy, aseptic meningitis, arthritis, acral paresthesias or dysesthesias (from peripheral neuropathy), or cognitive dysfunction (from CNS involvement) may be diagnostically useful.
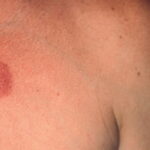
- Patients with borrelial lymphocytoma report a bluish red nodular swelling that is almost always on the lobe of the ear or the areola of the nipple. This symptom can occur in both the early disseminated and the late phases of Lyme disease and is seen exclusively in European patients.
- Children usually have lesions on the earlobe, while adults, both men and women, most commonly have lesions on the breast. Occasionally, borrelial lymphocytoma lesions occur on the scrotum, nose, and extremities. Nipple lesions tend to be painful, possibly because of rubbing against clothing.
- Erythema migrans may occur simultaneously with borrelial lymphocytoma, which indicates that borrelial lymphocytoma can occur very early in the disease course.
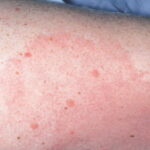
- Patients with acrodermatitis chronica atrophicans report a rash that is acral in distribution. Initially, discoloration and inflammation of the skin are seen, and later, severe atrophy is noted.
- Acrodermatitis chronica atrophicans tends to occur more commonly in females, especially older patients. Unlike erythema migrans, acrodermatitis chronica atrophicans tends to occur acrally, especially on the dorsal surfaces of the hands, feet, knees, and elbows. Early on, a minimally symptomatic erythema tends to occur in these locations.
- Progression is gradual, over months to years, from the inflammatory stage to a later atrophic stage. The latter stage is marked by a tendency towards central progression of the lesions, and more than 1 extremity may become involved. Eventually, subcutaneous fat is lost, and the skin becomes thin and wrinkled, similar to cigarette paper (see the image below).
- Some patients also report hypesthesia or dysesthesias characteristic of a polyneuropathy.
Physical
In many patients with early Lyme disease, physical examination alone is sufficient to establish the diagnosis of erythema migrans. Careful attention to the details often makes the difference between the need to proceed with further confirmatory tests and an empiric course of antibiotics. In particular, the examination must be interpreted in the epidemiologic context; this cannot be overemphasized. The season, geography, and a patient’s activities in those areas can be important diagnostic clues
A low-grade fever is not uncommon. High fever suggests another co-infecting tick-borne organism such as Ehrlichia or Babesia species or some other diagnosis altogether such as streptococcal cellulitis.
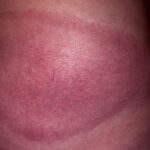
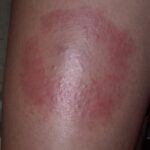
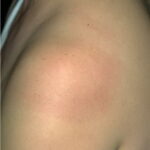
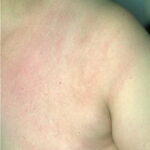
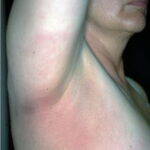
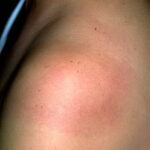
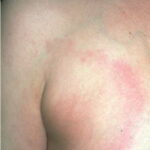
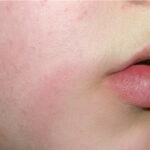
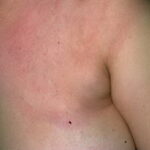
Classic erythema migrans begins as an erythematous macule or papule at the site of the tick bite (within 1-33 d; median is 7-10 d).7 Vesicles may be present. Often a central punctum is seen at the site of the bite. The eruption gradually expands over days to weeks (not hours or months), sometimes leaving central clearing. Note that this phenomenon, which has been termed a bull’s-eye or target lesion (see the image below) and was emphasized in the earlier literature, occurs only in a minority of patients (37% in one North American study). Central clearing and a more chronic course are more common in Europe, which accounts for the original European term erythema chronicum migrans.
Rashes very similar to erythema migrans have been reported in the southern United States from which B burgdorferi cannot be cultured. This disease is called southern tick-associated rash illness (STARI), or Master disease. As a group, distinctions can be made between classic erythema migrans and this illness, but significant overlaps exist such that the differences are not useful in diagnosing individual patients.
A number of important details are diagnostically useful. Note the following:
- Size: Erythema migrans varies in size (lesions are up to 70 cm; median is 16 cm). Some of the differential diagnoses (eg, bacterial cellulitis) become more unlikely with larger lesions, especially in a patient who does not appear ill. Although the CDC surveillance criteria for erythema migrans state that it must be greater than 5 cm in size, culture-proven cases have been occasionally documented that are smaller,8 and this size cut-off is only meant to be used for epidemiological purposes.
- Shape: Erythema migrans typically is round or oval and monocyclic. Occasionally, lesions can be triangular or linear, but this is seen less frequently.
- Associated symptoms: A paucity of pain or pruritus exists in most patients. When present, these localized symptoms tend to be mild. Associated systemic symptoms include low-grade fever, chills, fatigue, and neck stiffness. Very high fever or toxicity suggests an alternative diagnosis or an additional tick-borne infection such as babesiosis or ehrlichiosis.
- Location: Location of the bite is an important diagnostic clue. Ticks tend to feed in areas in which natural barriers prevent their forward progress, such as the popliteal fossa, groin, and axilla, or in areas in which elastic clothing or bra straps impede their journey. The thorax and trunk also are common spots. In children, the hairline and scalp are especially common locations.
- Evolution: Erythema migrans usually enlarges by a few centimeters per day and eventually fades within a few weeks, even without antibiotic treatment. Occasionally, the rash can be fleeting. Erythema migrans does not enlarge over hours, and it very rarely remains constant over weeks to months.
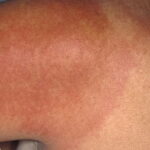
- Color and morphology: Most lesions are red . While central clearing has been emphasized in the past, the color of the lesion more commonly is uniform. Occasionally, the center is darker than the periphery. Lesions usually are flat but may be slightly raised (see the image below). Scaling does not usually occur. In some patients mistakenly treated with topical steroids, the rash may be quite pale.
- Other: Central clearing is more common in European patients with erythema migrans than in North American patients. Thus, while most lesions are flat erythemas, several variations are important to recognize, in particular, vesicular and centrally necrotic lesions.
Regional lymphadenopathy may be seen.
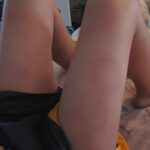
Early disseminated erythema migrans (multiple erythema migrans): Multiple erythema migrans lesions occur in approximately 20% of patients. Secondary lesions tend to be more uniform in morphology than the primary lesion. Necrosis and vesicles do not occur, nor does a central punctum. Since they are spread hematogenously, the locations of secondary lesions are not as restricted as in the primary lesion
Borrelial lymphocytoma usually occur on the earlobe or nipple, and the lesions are bluish-red nodules. The earlobe is the typical location in children (as is the scrotum), while the nipple location is more commonly seen in adults. Borrelial lymphocytoma tends to occur in areas of prior (or concurrent erythema migrans). The size is up to a few centimeters. Regional lymphadenopathy may be present (see the image below). Borrelial lymphocytoma is a form of B-cell pseudolymphoma. Other terms used to describe borrelial lymphocytoma include lymphadenosis benigna cutis, lymphocytoma cutis, cutaneous lymphoid hyperplasia, and Spiegler-Fendt lymphoid hyperplasia.
Acrodermatitis chronica atrophicans begins as an inflammatory phase marked by edema and erythema, usually on the distal extremities. At times, a faint bluish discoloration is found predominantly on extensor surfaces. The lesions have a predilection for the posterior heels and dorsal (extensor) surfaces of the hands, feet, elbows, and knees. Gradually, a central progression of the area involved occurs over months to years. The lesions also tend to become symmetric. The buttocks often become involved. Later, atrophy supervenes and thin cigarette-paper skin is seen. Because of the loss of subcutaneous fat, underlying venous structures are more visible, and the skin becomes thin, atrophic, and dry. Fibrous juxtaarticular nodules or bands may be seen on the extensor surfaces of the elbows and knees. Signs of peripheral neuropathy may coexist with acrodermatitis chronica atrophicans.
Other skin lesions have been associated with B burgdorferi infection, but whether they are part of the syndrome of Lyme disease is controversial. The lesion for which the most evidence of causality has been reported is morphea (localized scleroderma), which develops in roughly 10% of European patients with borrelial lymphocytoma and acrodermatitis chronica atrophicans. B burgdorferi has been isolated from these lesions but not from North American patients with morphea. Other European reports less commonly link lichen sclerosis et atrophicans, progressive facial hemiatrophy (Parry-Romberg syndrome), and eosinophilic fasciitis with B burgdorferi infection
Causes
The cause of Lyme disease is infection by the spirochete, B burgdorferi. This organism, whose complete genome was described in 1998, has several distinct genetic groupings; as well, there is evidence that additional strains or closely related Borrelia species also exist.
The generic species is B burgdorferi (eg, sensu lato). Within this species exists several well-characterized groupings that account for the different clinical manifestations seen in North America and Europe. Three groups are well established, including B burgdorferi sensu stricto, Borrelia garinii, and Borrelia afzelii. Many other strains exist, but most are not pathogenic to humans. This is an area of active and constantly evolving research. Note the following:
- B burgdorferi sensu stricto is the strain that constitutes all North American isolates and is found in Europe as well. In European erythema migrans, B afzelii can be isolated from about 80% of lesions and B garinii from 15%.9
- B garinii, found exclusively in Europe, has some neurotropism and is the isolate that accounts for most cases of the neurologic syndrome lymphocytic meningoradiculitis (Bannwarth syndrome) and a white matter encephalitis, which is rare in North America. However, this organism does cause all the various cutaneous manifestations described above.
- Although B afzelii is the most common organism causing acrodermatitis chronica atrophicans, all 3 groups have been isolated from these patients.
The primary risk factor for developing cutaneous manifestations of Lyme disease is exposure to Ixodes ticks.
Laboratory Studies
In most patients with erythema migrans (EM), a carefully elicited history (including definitions of epidemiologic context) and a physical examination are all that is required to establish the diagnosis. The US Centers for Disease Control and Prevention (CDC) accepts physician-diagnosed erythema migrans of greater than 5 cm in size in its case definition. (In clinical practice, erythema migrans can be smaller than this size. The 5-cm limit was designed for surveillance purposes.) No tests, including serologic tests for Lyme disease, are indicated in these patients. CBC counts and erythrocyte sedimentation rates often are normal. Antibody titers B burgdorferi may be falsely negative at this early stage of disease and should not affect the decision to treat.
In patients in whom significant ambiguity exists concerning the diagnosis after the history and physical examination are performed, several strategies can be used.
- Strategy one: Empiric antibiotic therapy is reasonable if erythema migrans is moderately probable. Therapy using amoxicillin or doxycycline usually is safe and inexpensive and can be initiated immediately. If the lesion is erythema migrans, prompt improvement (within a few days) and resolution is expected.
- Strategy two: Acute and convalescent-phase serologic testing has some drawbacks. A waiting period of several weeks may be required before antibodies appear in the serum in sufficient quantities to measure; therefore, early false seronegative results are common. In addition, the notion of waiting before treatment is unappealing to many patients as there is risk that easy-to-treat localized disease will disseminate (and potentially become more difficult to treat). Physicians must understand the limitations of serologic tests.
- The CDC currently recommends a 2-step testing procedure consisting of a screening enzyme-linked immunoassay (ELISA) or immunofluorescent assay followed by a confirmatory Western immunoblot test on any samples with positive or equivocal results on ELISA.
- Newer serological testing with the C6-peptide and VslE are promising new tests. While they are still not always routinely available, some commercially available C6 assays have been developed. Importantly, remember, however, that whichever serological test is performed, the principle of timing (see below) is still operant.10,11
- Timing is important, since many patients at the erythema migrans stage are seronegative. In addition, numerous causes of false-positive ELISA test results exist, such as various infectious and immunologic diseases. Patients with past Lyme disease may be persistently seropositive. All previously vaccinated patients have positive ELISA test results.
- For all these reasons, sequential serologic testing has serious limitations for use in individual patients with early Lyme disease, and its routine use should be discouraged.
- Strategy three: Perform skin biopsy for histology and culture or polymerase chain reaction (PCR) of the rash to obtain further diagnostic information.
- Histologic analysis of skin biopsy specimens (see Histologic Findings) is not diagnostic for erythema migrans, and spirochetes are seen uncommonly. In some circumstances, biopsy can help exclude other diagnoses but does not include erythema migrans.
- Culture of punch biopsy material is another method. While the organism has fastidious growth requirements, culture in the appropriate medium (BSK-II) is positive in approximately 75% of patients. This test is not universally available, but in patients with ambiguous disease, it may be useful in establishing the diagnosis.
- PCR is growing in uses and availability. Nevertheless, as of 2005, PCR remains a research technique, in part because labs performing PCR tests must be meticulous in technique to minimize the likelihood of false-positive results. Most importantly, it is not readily available to most clinicians in routine practice. In addition, no large clinical series have been reported that assess the performance of the test in the nonresearch setting.
- Strategy four: Observing the spontaneous evolution of the rash over several days is safe and universally available. In most patients with erythema migrans, some expansion of the rash is expected over 2-3 days without antibiotics. In patients in whom disease is ambiguous, especially when culture and PCR are unavailable, this is a reasonable alternative to immediate empiric therapy.
Most, but not all, patients with borrelial lymphocytoma are seropositive for antiborrelial antibodies. This is true for all early disseminated manifestations of Lyme disease.
In the typical patient, no tests beyond serologic testing are necessary. When the clinical syndrome is not classic (borrelial lymphocytoma [BL] lesion not found on earlobe or breast, seronegative patients), biopsy may be indicated. Histopathology shows dermal lymphocytic infiltration with germinal centers, which is suggestive but not diagnostic. The organism rarely may be cultured from lesions.
Essentially all patients with acrodermatitis chronica atrophicans (ACA) are seropositive for antiborrelial antibodies. Seriously question the diagnosis in seronegative patients.
Results of some laboratory studies may suggest some of the other co-infecting tick-borne pathogens such as ehrlichial or babesial species. Most patients with ehrlichiosis have elevated levels of hepatic transaminases, leukopenia, and/or thrombocytopenia. In addition, some patients have morulae (intracytoplasmic inclusions) in white blood cells as demonstrated on peripheral blood smears. Patients with babesiosis often are anemic (hemolytic type) and may have thrombocytopenia. Blood smears reveal the malarialike intraerythrocytic parasite in this disease as well.
Histologic Findings
Histologic findings in erythema migrans are nonspecific, usually showing a perivascular cellular infiltrate consisting of lymphocytes, plasma cells, and histiocytes. Occasionally, mast cells and neutrophils are seen. Central biopsies may show eosinophilic infiltrate consistent with a local reaction to an arthropod bite. Spirochetes occasionally may be identified using silver or antibody-labeled stains, although usually, a paucity of spirochetes is found in the tissues of patients with Lyme disease. In addition, cultures for B burgdorferi are positive in approximately 75% of patients.
Borrelial lymphocytoma biopsy specimens show a dense dermal lymphocytic infiltrate with lymphoid follicles and pseudogerminal centers. Lymphocytes with both B-cell and T-cell markers, occasional macrophages, plasma cells, and eosinophils are seen.
In acrodermatitis chronica atrophicans, biopsy specimens from early lesions show a lymphocytic dermal infiltrate, sometimes perivascular in location, with some vascular telangiectasia and lymphedema. Plasma cells also may be seen in the cellular infiltrate. Later lesions demonstrate epidermal thinning with loss of skin appendages. At this stage, plasma cells may be the only feature to distinguish acrodermatitis chronica atrophicans from morphea. The fibrotic nodules show fibrosis of the deeper dermis and sometimes, hyalinization of collagen bundles. B burgdorferi occasionally can be cultivated from the lesions; in 1 patient, cultivation was successful more than 10 years after the lesion’s first appearance
Medical Care
Cutaneous manifestations of Lyme disease are treated using antibiotics. Important considerations include the stage of the disease, presence of associated neurologic symptoms or signs (especially in borrelial lymphocytoma [BL] and acrodermatitis chronica atrophicans [ACA]), and patient factors (duration of symptoms, allergies, age, pregnancy status).
Controversies in treatment and diagnostic entities
Controversy regarding the treatment of Lyme disease abounds, including an antitrust investigation by the Connecticut Attorney General into the development process for the Lyme disease treatment guidelines from the Infectious Disease Society of America. The Attorney General claimed the process was tainted by suppression of scientific evidence and conflicts of interest.12
Controversy also surrounds many aspects of the diagnosis and treatment of Lyme disease. Some have proposed existence of an “Axis of Evil,” which would include the Internet, for promoting Lyme hysteria; particular specialty laboratories, for allegedly performing inaccurate testing; and physicians, specifically those who prescribe prolonged and unnecessary courses of antibiotic treatment.13
The existence and treatment of conditions termed chronic Lyme disease and posttreatment Lyme disease have been called into question as a result of a lack of direct evidence of persistent infection.14,15 Extended antibiotic therapy, sometimes longer than 6 months, has been advocated for these poorly defined conditions, which not only can cause great harm to patients but has resulted one or more deaths.16 Hassett et al report associated psychiatric comorbidity in patients with chronic Lyme disease.17
In 2006, the guidelines committee of the Infectious Disease Society of America stated that a group of symptoms termed “persistent Lyme disease symptoms” (eg, headaches, mood disturbances, fatigue, poor memory, joint pain) are merely the “aches and pains of daily living.” Further, an ad hoc international Lyme group stated they are “symptoms common in persons who have never had Lyme disease.” Cameron has proposed that despite the controversy surrounding persistent Lyme disease symptoms, evidence to deny care is lacking and to do so is neglectful to patient care.
Medication
In general, skin manifestations of Lyme disease respond promptly to appropriate antibiotic therapy. Early manifestations respond more rapidly than later manifestations. Data regarding the best route and duration of antibiotic therapy are evolving and are, to some extent, controversial. Some researchers claim an abundance of overtreatment and overdiagnosis of Lyme disease, pointing to several nonspecific conditions and symptoms that have been linked to Lyme disease. They indicate that if these sequelae remain after appropriate therapy, other causes must be carefully and conclusively excluded.20
For solitary erythema migrans, oral antibiotics clearly provide effective therapy. The duration of recommended therapy ranges from 10-30 days. In the past, this author has recommended 3 weeks of treatment, since a sizable minority of patients with solitary erythema migrans have evidence of hematogenous dissemination, even in the absence of symptoms. In addition, strong evidence indicates that patients with early disseminated Lyme disease have equally good outcomes after 3 weeks of oral antibiotics compared with 2 weeks of parenteral therapy. Therefore, patients with erythema migrans and asymptomatic disseminated disease are treated adequately using the 3-week course.
While the approach described above has inherent logic, newer studies based on actual outcomes have called into question duration of therapy beyond 10 days. In one retrospective study of 607 patients, 25% of whom had early disseminated disease, outcomes of patients treated for 10 days were equivalent to those treated with longer courses.21 Based on this, and previous studies, many authorities consider 2 weeks of antibiotics to be adequate therapy for erythema migrans and some use as few as 10 days.
For pregnant women with erythema migrans, some physicians recommend parenteral therapy, although data on this are limited. Isolated reports exist of transplacental transmission from the mother to fetus. One European descriptive study showed good results of parenteral ceftriaxone in pregnant women with erythema migrans.22
Borrelial lymphocytoma usually is treated with 14-21 days of oral antibiotics, but when symptoms of dissemination are noted, parenteral therapy sometimes is used. Borrelial lymphocytoma is sufficiently uncommon that no comparative trials address the ideal duration of treatment, route of administration of the antibiotic, or the choice of medication. For the same reasons as in erythema migrans, some logic exists in using a 3-week course of antibiotics.
Acrodermatitis chronica atrophicans usually is treated with 1-month course of oral antibiotics, usually a beta-lactam or doxycycline. One study showed fewer relapses with 30 days compared to 20 or fewer days of therapy. In the same study, 30 days of oral antibiotics were more effective than 15 days of IV ceftriaxone (2 g/d).23 Ensure that no neurologic manifestations are present before embarking on oral therapy.
Antibiotics
Directed against B burgdorferi sensu lato. They are indicated for all the cutaneous manifestations of Lyme disease.
Amoxicillin (Amoxil, Dispermox, Trimox)
DOC for pediatric patients <9 y and may be used in pregnancy (assuming parenteral antibiotics are not used). Does not treat disease from co-infecting organisms (eg, ehrlichiosis, babesiosis).
Adult
250-500 mg PO tid; not to exceed 3 g/d
Pediatric
20-50 mg/kg/d PO divided tid; not to exceed 3 g/d
Probenecid (Benemid) increases effects
Documented hypersensitivity
Pregnancy
B – Fetal risk not confirmed in studies in humans but has been shown in some studies in animals
Precautions
Adjust dose in renal impairment
Doxycycline (Vibramycin)
DOC for adults with erythema migrans. Inhibits protein synthesis and, thus, bacterial growth by binding to 30S and possibly 50S ribosomal subunits of susceptible bacteria. Advantage is that it covers co-infecting ehrlichial and rickettsial species.
Dosing schedule aids in compliance.
Adult
100 mg PO bid
Pediatric
<8 years: Not recommended
>8 years: 2-5 mg/kg PO qd or divided bid; not to exceed 200 mg/d
Bioavailability decreases with antacids containing aluminum, calcium, magnesium, iron, or bismuth subsalicylate; tetracyclines can increase hypoprothrombinemic effects of anticoagulants.
- Documented hypersensitivity; severe hepatic dysfunction
Pregnancy
D – Fetal risk shown in humans; use only if benefits outweigh risk to fetus
Precautions
Photosensitivity may occur with prolonged exposure to sunlight or tanning equipment; reduce dose in renal impairment; consider drug serum level determinations in prolonged therapy; tetracycline use during tooth development (last one half of pregnancy through age 8 y) can cause permanent discoloration of teeth; Fanconilike syndrome may occur with outdated tetracyclines
Cefuroxime (Ceftin)
Second-generation cephalosporin maintains the gram-positive activity of first-generation cephalosporins. Has explicit FDA approval for erythema migrans. For cost reasons, it is an alternate drug.
In cases where bacterial cellulitis cannot be confidently distinguished from erythema migrans, this drug treats both processes.
Adult
500 mg PO bid
Pediatric
Children: 250 mg PO bid for 20 d
Adolescents: Administer as in adults
Disulfiram-like reactions may occur when alcohol is consumed within 72 h after taking cefuroxime; may increase hypoprothrombinemic effects of anticoagulants; may increase nephrotoxicity in patients receiving potent diuretics such as loop diuretics; coadministration with aminoglycosides increases nephrotoxic potential
Documented hypersensitivity
Pregnancy
C – Fetal risk revealed in studies in animals but not established or not studied in humans; may use if benefits outweigh risk to fetus
Precautions
Administer one-half dose if CrCl level is 10-30 mL/min and one-quarter dose if <10 mL/min (high doses may cause CNS toxicity); bacterial or fungal overgrowth of nonsusceptible organisms may occur with prolonged or repeated therapy
Erythromycin (E-Mycin, Eryc, Ery-Tab)
Inhibits bacterial growth, possibly by blocking dissociation of peptidyl tRNA from ribosomes arresting RNA-dependent protein synthesis. For treatment of staphylococcal and streptococcal infections.
In clinical studies, did not perform as well as other oral antibiotics despite good in vitro sensitivities. Not recommended except in unusual circumstances (eg, pregnant patient with severe allergy to beta-lactam antibiotics).
Adult
250 mg erythromycin stearate/base (or 400 mg ethylsuccinate) PO q6h, or 500 mg q12h (1 h ac or 2 pc)
Alternatively, 333 mg PO q8h; increase to 4 g/d depending on severity of infection
Pediatric
30-50 mg/kg/d (15-25 mg/lb/d) PO divided q6-8h; double dose for severe infection
Coadministration may increase toxicity of theophylline, digoxin, carbamazepine, and cyclosporine; may potentiate anticoagulant effects of warfarin; coadministration with lovastatin and simvastatin increases risk of rhabdomyolysis
Documented hypersensitivity; hepatic impairment
Pregnancy
B – Fetal risk not confirmed in studies in humans but has been shown in some studies in animals
Precautions
Caution in liver disease; estolate formulation may cause cholestatic jaundice; GI tract adverse effects are common (administer doses pc); discontinue if nausea, vomiting, malaise, abdominal colic, or fever occur
Azithromycin (Zithromax)
Second-line drug. Like erythromycin, has excellent in vitro sensitivities but has underperformed compared to amoxicillin in 1 large study. Conversely, in several European studies, has been shown to be equal to beta-lactam and tetracycline group antibiotics. Because of once-daily dosing, should be considered in pregnant patients who are allergic to beta-lactams and in patients where compliance is a major issue.
Adult
500 mg PO qd for 5 d, followed by 5-day hiatus, then second course of 500 mg PO qd for 5 d
Pediatric
10 mg/kg PO once on day 1; not to exceed 500 mg/d, followed by 5 mg/kg PO on days 2-5; not to exceed 250 mg/d
Alternatively, 5-12 mg/kg/d PO for 15 d
May increase toxicity of theophylline, warfarin, and digoxin; effects are reduced with coadministration of aluminum and/or magnesium antacids; nephrotoxicity and neurotoxicity may occur when coadministered with cyclosporine
Documented hypersensitivity; hepatic impairment; do not administer with pimozide
Pregnancy
B – Fetal risk not confirmed in studies in humans but has been shown in some studies in animals
Precautions
Site reactions can occur with IV route; bacterial or fungal overgrowth may result with prolonged antibiotic use; may increase hepatic enzymes and cholestatic jaundice; caution in patients with impaired hepatic function, prolonged QT intervals, or pneumonia; caution in patients who are hospitalized, geriatric, or debilitated