Epidermodysplasia verruciformis
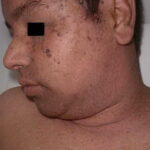
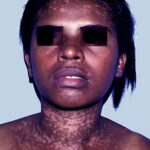
Epidermodysplasia verruciformis (EV) is a rare, inherited disorder that predisposes patients to widespread human papillomavirus (HPV) infection and cutaneous squamous cell carcinomas.1,2 Although epidermodysplasia verruciformis is most commonly inherited in an autosomal recessive manner, sporadic, sex-linked, and autosomal dominant inheritance have been described. The disease is characterized by chronic infection with HPV. Widespread skin eruptions of flat-to-papillomatous, wartlike lesions and reddish-brown pigmented plaques on the trunk, the hands, the upper and lower extremities, and the face are typical.
Malignant skin tumors (carcinomas), especially squamous cell carcinoma (in situ or invasive), develop frequently in these patients (30–70%), most commonly in sun-exposed areas starting between the ages of 20 and 40 years. Skin cancers initially appear on sun-exposed areas, such as the face, neck, chest, and arms. Patients with epidermodysplasia verruciformis are usually infected with multiple types of HPV, including common types that affect individuals without epidermodysplasia verruciformis (eg, HPV type 3 and 10) and those unique to epidermodysplasia verruciformis, the so called epidermodysplasia verruciformis–associated HPVs (EV-HPVs).
More than 30 EV-HPVs, such as types 4, 5a, 5b, 8,9, 12, 14, 15, 17, 19-25, 36-38, 47, and 50, have been identified in epidermodysplasia verruciformis lesions. Some EV-HPVs are detected in up to 20% of the non–epidermodysplasia verruciformis population, but they are only pathogenic in epidermodysplasia verruciformis patients. HPV-5 and HPV-8 have been isolated in more than 90% of epidermodysplasia verruciformis–associated squamous cell carcinomas.
Pathophysiology
The pathophysiology of epidermodysplasia verruciformis is linked to defective cell-mediated immunity, with elucidation of mutations in EVER1 and EVER2 genes (band 17q25).2,3 Their gene products are integral membrane proteins localized to the endoplasmic reticulum.
Although the role of EVER1 and EVER2 genes in the pathogenesis of epidermodysplasia verruciformis remains unclear, one hypothesis is that they are involved in the control of HPV infection within keratinocytes, or they play a role in the immune response to the infection itself. Intracellular zinc homeostasis regulated by a complex of EVER proteins and zinc transporter proteins may play a role in inhibiting EV-HPV expression.4 However, an estimated 25% of patients with epidermodysplasia verruciformis lack mutations in EVER1 and EVER2, with the genetic defect in these patients not yet elucidated.5
Several epidermodysplasia verruciformis variants have been described, and the majority of these cases occur in association with immunosuppression, such as HIV infection, organ transplantation, or idiopathic lymphopenia. Zavattaro et al reported a rare case of an epidermodysplasia verruciformis patient who had clinical features of epidermodysplasia verruciformis but lacked the EVER1 or EVER2 mutation.6 This patient was older at diagnosis and had no premalignant or malignant lesions upon examination. Defective Fas protein function (CD95, apoptosis receptor) was identified along with perforin gene variations, suggesting that this combination resulted in increased susceptibility to HPV infection owing to defective viral clearance.
In addition, a profound CD8+ T-cell lymphocytopenia was identified, a finding also described by Azzimonti et al in a patient who also had a clinical diagnosis of epidermodysplasia verruciformis but who lacked EVER1 or EVER2 mutations.7
The papillomavirus genus is a member of the Papovaviridae family. HPVs are small, nonenveloped viruses, measuring approximately 55 nm in diameter. Their icosahedral capsid is composed of 72 capsomers, with a 56,000-d major protein, which is the genus-specific antigenic determinant of the virus, and a 76,000-d minor protein. The HPV genome contains a double-stranded circular DNA of approximately 7900 base pairs, functionally divided into an early region (E) of 5-7 open reading frames E1-E7, a late region (L) of open reading frames L1 and L2, and a noncoding upstream regulatory region. The HPV types are primarily classified on the basis of their DNA homology.
Patients with epidermodysplasia verruciformis have a defective cell-mediated immune response to HPV infection. Many HPV types found in epidermodysplasia verruciformis lesions are nonpathogenic to the general population. The exact mechanism by which cancer occurs frequently in patients with epidermodysplasia verruciformis is unclear. The role of HPV in cancer development is supported by the identification of viral DNA within epidermodysplasia verruciformis–induced malignancies. Carcinogenic cofactors, such as ultraviolet B and x-ray irradiation, are probably involved in the progression from benign warts (verrucae) to cancer. Cells with early signs of malignant transformation have been found closely connected with virus-infected epidermal regions.
The exact mechanisms involved in the malignant transformation of keratinocytes in skin lesions of patients with epidermodysplasia verruciformis are still unclear. Studies have shown that interactions occur between oncogenic HPVs and antioncogene proteins, such as p53 and pRb, in cell cycle regulation, DNA repair, and the execution of programmed cell death (apoptosis).
The persistence of HPV infection in epidermodysplasia verruciformis is thought to be the result of an immunogenetic defect, which generates several cytokines capable of down-regulating cell-mediated immunity. Patients with epidermodysplasia verruciformis reportedly show an increased rate of low-production genotypes of interleukin 10 compared with control subjects. Patients with epidermodysplasia verruciformis and skin cancer are more likely to have low-production interleukin 10 genotypes than patients with benign forms of epidermodysplasia verruciformis.8
In epidermodysplasia verruciformis tumors, gene products transcripts of E6 and E7 (the early region of viral genes) are detected. Within the early region of the HPV genome, E6 and E7 code for major oncoproteins responsible for the oncogenic potential of HPV. These viral proteins are crucial for tumorigenesis. In cancerous lesions, the high-risk HPV types, such as HPV types 5, 8, and 47, selectively retain and express the E6 and E7 portions of the viral genome. Working together, these E6/E7 regions cause cell immortalization, or failure of programmed cell death, resulting in transformation of normal human keratinocytes into malignant cells.9
Both E6 and E7 are multifunctional proteins that promote cell growth via multiple mechanisms. Each has the ability to neutralize an antioncogene product, specifically p53 and pRb, that is essential for intracellular defense mechanisms against the development of neoplasms. However, the exact mechanism of carcinogenesis of E6 and E7 oncoproteins and the effects of these oncoproteins on p53 and pRb are unclear.
Failure of programmed cell death to eliminate cells with DNA damage may play an important role in malignant transformation of squamous epithelium. A decrease in UV-induced DNA repair synthesis, coupled with an oncogenic viral infection, further enhances the susceptibility toward somatic mutations and malignant transformation in patients with epidermodysplasia verruciformis.10,11,12,13
Renal transplant recipients and immunosuppressed patients have an increased risk of developing lesions of epidermodysplasia verruciformis.5
History
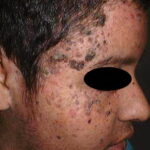
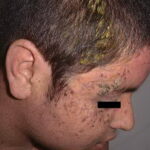
- Epidermodysplasia verruciformis usually begins in infancy or early childhood, with the development of various types of flat, wartlike lesions and confluent plaques on the skin, especially on dorsal hands, extremities, face, and neck.
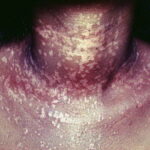
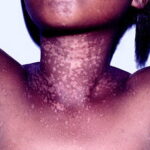
- Patients may also develop tinea versicolor -like lesions on the trunk.
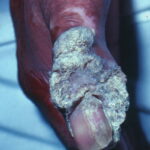
- Epidermodysplasia verruciformis lesions may progress to form verrucous plaques and nodules, or they may transform into invasive squamous cell carcinomas, most commonly between the ages of 20 and 40 years.
- The clinical course of epidermodysplasia verruciformis is protracted. As the disease progresses, some lesions disappear, while new lesions may appear on other areas of the body. The rate of appearance of new lesions varies considerably.
- The diagnosis of epidermodysplasia verruciformis should be suspected in the clinical setting of numerous verrucous lesions or when lesions are resistant to appropriate therapy.
- Epidermodysplasia verruciformis variants may be suspected when a patient has the typical clinical presentation in the setting of epidermodysplasia verruciformis–associated HPV but lacks nonmelanoma skin cancers at a young age or has late-onset disease.
Physical
- Pertinent physical findings are limited to the skin and rarely occur on the mucosa.
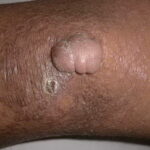
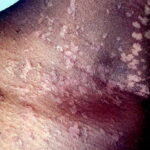
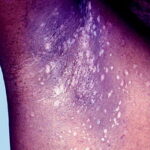
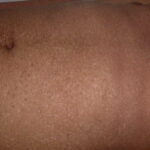
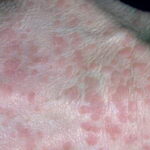
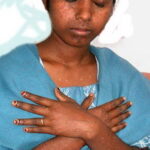
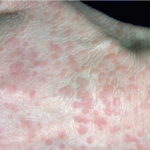
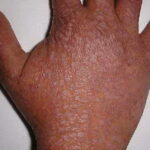
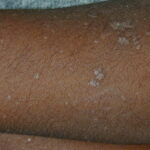
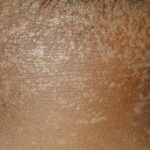
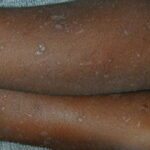
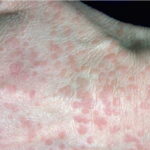
- Primary skin lesions manifest as 2 types, although they generally are polymorphic. The first type is flat, wartlike lesions resembling verruca plana; they are flat-topped papules with scaly, hyperpigmented or hypopigmented, sometimes confluent patches or plaques. Flat macules and reddish brown plaques with slightly scaly surfaces and irregular borders are also noted, as shown in the image below. These lesions may resemble tinea versicolor. Papules on the knees, the elbows, and the trunk may coalesce into large plaques
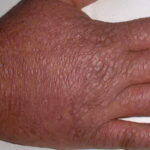
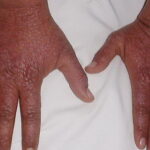
The second type is verrucous or seborrheic keratosis –like lesions; they are commonly seen on sun-exposed skin, including dorsum of hands
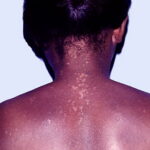
- The wartlike lesions are mostly localized on sun-exposed areas, mainly distributed on the hands, the feet, and the face, sometimes in a linear arrangement, as demonstrated in the image below. The pigmented plaques preferentially involve the trunk, the neck, and the proximal extremities. The lesions may be found on the palms and the soles, in the axillae, and on the external genitalia. The mucous membranes (conjunctiva and oral cavity) are rarely affected.
- Cutaneous lesions induced by EV-HPVs vary from flesh-colored warts (verruca vulgaris) to red, reddish-brown, and brown plaques.
Causes
- Epidermodysplasia verruciformis–associated HPVs can be divided into 2 groups.
- One group has high oncogenic potential (HPV types 5, 8, 10, and 47). More than 90% of epidermodysplasia verruciformis–associated skin cancers contain these virus types.
- The other group has low oncogenic potential (HPV types 14, 20, 21, and 25). These types are usually detected in benign skin lesions.
- Proposed mechanisms for the development of epidermodysplasia verruciformis include the following:
- An autosomal recessive mode of inheritance is supported by the finding that 10% of patients with epidermodysplasia verruciformis are offspring of consanguineous marriages. X-linked inheritance has rarely been reported.1 A clear mode of inheritance is not evident in all cases.
- Pathogenic mutations in 2 adjacent genes, EVER1 and EVER2, have been identified.2,3
- Major histocompatibility complex (MHC) class II alleles (DR-DQ) have been found in a large series of patients with epidermodysplasia verruciformis from Europe, Africa, and America.
- Neither chromosomal abnormalities nor the relationship to any specific MHC class I antigens has been found in patients with epidermodysplasia verruciformis.
- The exact mechanisms involved in the keratinocytic transformation within epidermodysplasia verruciformis skin lesions are unclear. Transcripts of the early region of viral genomes (E6 and E7 gene proteins) have been detected in epidermodysplasia verruciformis tumors. However, in most carcinomas, viral sequences are not integrated into the host genome.
- Studies have shown that interactions occur between oncogenic HPVs and the antioncogene products, p53 and pRb, in cell cycle regulation, DNA repair, and the execution of programmed cell death (apoptosis). Failure of programmed cell death to eliminate cells with DNA damage may play an important role in the malignant transformation of squamous epithelium, with resultant proliferation, disruption of epithelial structural order, and development of cellular atypia. A decrease in UV-induced DNA repair synthesis, coupled with antioncogenic viral infection, further enhances the disposition for somatic mutations and malignant transformation in patients with epidermodysplasia verruciformis.
- A specific defect of cell-mediated immunity, manifested by the inhibition of natural cytotoxicity and the proliferation of T lymphocytes against HPV-infected squamous cells in epidermodysplasia verruciformis skin lesions, is a characteristic feature of epidermodysplasia verruciformis.
- Chronic sun-exposure coupled with immunologic defects in patients with epidermodysplasia verruciformis is likely to induce mutations of the tumor suppressor gene protein (p53), leading to the development of malignant skin cancer in adult patients.
- UV-B–induced local immunosuppression on the skin of patients with epidermodysplasia verruciformis is known to be related to overproduction of immunosuppressive cytokines, such as tumor necrosis factor-alpha (TNF-a), transforming growth factor-beta (TGF-b), interleukin 4, and interleukin 10, as well as excessive formation of cis- urocanic acid.
- Studies have implicated a defect within keratinocytes. The activity of Langerhans cell antigen presentation appears normal in epidermodysplasia verruciformis, thus suggesting other cells cause immunotolerance to epidermodysplasia verruciformis–associated HPVs.
- Lesions of epidermodysplasia verruciformis have been associated with common variable immunodeficiency and graft versus host disease.6
Laboratory Studies
- Human papillomaviruses (HPVs) can be detected by in situ hybridization, using anti-HPV antibodies on formalin-fixed skin tissue specimens. Unfortunately, this viral typing is not readily available on a commercial basis. Typing can also be performed by polymerase chain reaction on fresh or frozen tissue specimens.15
- EVER1 and EVER2 mutational analysis can be evaluated using single-stranded conformational polymorphism analysis, to screen polymerase chain reaction fragments specific to exons for mutation(s) of the EVER1 and EVER2 genes, in a DNA sample from leukocytes.
- Defective cell-mediated immunity can be detected by the following studies:
- Keratinocytes isolated from premalignant lesions of patients with epidermodysplasia verruciformis with HPV type 5 genomes show inhibition to natural cell-mediated cytotoxicity by normal peripheral blood mononuclear cells, whereas normal keratinocytes do not.
- Patients with mixed HPV or HPV type 3 infection may demonstrate cutaneous anergy to locally applied contact sensitizers, such as dinitrochlorobenzene.
- A normal number of antigen-presenting Langerhans cells are found in patients with epidermodysplasia verruciformis. However, the possibility of a genetically determined defective functionality of these cells, leading to abnormal presentation and recognition of HPV antigens, has been considered.
Histologic Findings
Biopsy is performed for early detection of premalignant and malignant lesions and for the identification of epidermodysplasia verruciformis–associated HPVs. The most characteristic findings are seen within the epidermis. The classic histologic manifestation of epidermodysplasia verruciformis is a verruca plana–like lesion with mild hyperkeratosis and acanthosis, in which the keratinocytes contain perinuclear halos and blue-gray pallor, as is demonstrated in the image below. Perinuclear halos are a specific cytopathic effect, that is, clear cells in the granular and spinous layers with occasional enlarged, hyperchromatic, atypical nuclei, are present
The nucleoplasm is clear, and keratohyalin granules of various sizes and shapes are present. The keratin layer is loose, with a basket weave–like appearance. In premalignant tumors, the normal keratinocyte maturation is preserved. In contrast, in malignant lesions, the normal surface maturation of keratinocytes is lost. HPV can be detected in infected keratinocyte nuclei by in situ hybridization, particularly in the upper layers of the epidermis
Premalignant lesions display features similar to actinic keratosis, with prominent atypical, dyskeratotic cells. The cytopathic effects of viral warts are often absent, although large amounts of HPV DNA can be detected, as is shown in the image by using immunostains on a skin lesion
Invasive malignant tumors most commonly show squamous and occasionally adnexal differentiation
Treatment
Medical Care
No therapy for epidermodysplasia verruciformis is definitive. Treatment of epidermodysplasia verruciformis includes preventive measures, the most important of which is strict sun avoidance and protection, beginning as soon as the diagnosis is made.
- Nonsurgical therapies for the management of skin cancers include topical imiquimod and 5-fluorouracil, systemic retinoids, interferon, and 5-aminolevulinic acid photodynamic therapy.17,18
- In advanced human papillomavirus (HPV)–related carcinomas, an experimental therapy involves treatment with a combination of 13-cis retinoic acid and interferon alpha or cholecalciferol analogues.19
- For localized multiple malignant lesions, autotransplantation of skin from uninvolved areas has been reported with success in preventing further development of cancers.
- UV-B exposure, UV-A exposure, and x-ray irradiation should be avoided because radiation therapy often promotes the recurrence of more aggressive skin cancers.
Surgical Care
Surgical and electrosurgical removal and cryotherapy are used in the treatment of benign and premalignant skin lesions. Surgery is also indicated for treatment of malignant lesions. If skin grafting is necessary, the graft should be from sun-protected skin.
- Epidermodysplasia verruciformis–associated HPVs can be divided into 2 groups.