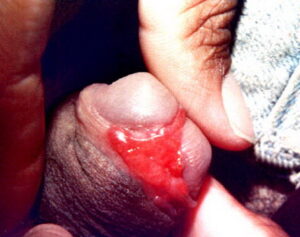
Donovanosis
Granuloma inguinale is a chronic bacterial infection that frequently is associated with other sexually transmitted diseases. Granuloma inguinale is characterized by intracellular inclusions in macrophages referred to as Donovan bodies. Granuloma inguinale usually affects the skin and mucous membranes in the genital region, where it results in nodular lesions that evolve into ulcers. The ulcers progressively expand and are locally destructive
The intracellular organism responsible for granuloma inguinale was initially described by Donovan over a century ago, and subsequently, the bacterium was classified in 1913 as Calymmatobacterium granulomatis. Although Anderson suggested that the organism be eponymously named Donovania granulomatosis, Carter et al discovered that the molecular structure of the causative organism was similar to Klebsiella species and reclassified the gram-negative pleomorphic bacillus as Klebsiella granulomatis.1,2
The mode of transmission of granuloma inguinale primarily occurs through sexual contact; however, it is hypothesized to have low infectious capabilities because repeated exposure is necessary for clinical infection to occur. Additionally, granuloma inguinale may also be obtained through the fecal route or by passage through an infected birth canal.
History
Although the exact incubation period for granuloma inguinale is unknown, it ranges from a day to a year, with the median time being 50 days.3
Physical
Morphology
The 4 main types of cutaneous lesions are as follows:
- Nodular: The initial granuloma inguinale lesion is a papule or nodule that arises at the site of inoculation. The nodule is soft, often pruritic and erythematous, and eventually ulcerates. A nodule may be mistaken for a lymph node [ie, pseudobubo].
- Ulcerovegetative (most common): These granuloma inguinale lesions develop from nodular lesions and consist of large, usually painless, expanding, suppurative ulcers. The ulcers have clean, friable bases with distinct, raised, rolled margins and have a tendency to bleed easily. The ulcers are “beefy red” and slowly expand centrifugally, eventually becoming more granulomatous with serpiginous borders. They are commonly located in the skin folds, and autoinoculation is a common feature, resulting in lesions on adjacent skin. Ulcers often become secondarily infected with other types of bacteria and emit a putrid odor.
- Cicatricial: Dry ulcers evolve into cicatricial plaques and may be associated with lymphedema.
- Hypertrophic or verrucous (relatively rare): This proliferative reaction, with the formation of large vegetating masses, may resemble genital warts.
Elephantiasislike swelling of the external genitalia is a frequent complication and is found most often in infected females in the late stage of granuloma inguinale.
- The most common locations of granuloma inguinale lesions in men are the sulcocoronal and balanopreputial regions, as well as the anus.
- In women, granuloma inguinale lesions occur on the labia minora, the mons veneris, the fourchette, and/or the cervix. Cervical involvement occurs in 10% of cases.
- Children are frequently infected via contact with an adult; however, this is not necessarily the result of sexual abuse.3
Extragenital involvement
- Extragenital involvement occurs in 6% of granuloma inguinale cases.
- Autoinoculation or direct extension may lead to involvement of the lips, oral/gastrointestinal mucosa, scalp, abdomen, arms, legs, and bones.
- Lymphadenopathy does not occur as a result of the primary infection with Klebsiella granulomatis, but, rather, it occurs from secondary bacterial infections. Pseudobuboes resemble lymph nodes, but they are just nodular lesions.
- Disseminated lesions associated with systemic symptoms are frequently reported in endemic regions.3 Hematogenous dissemination to the spleen, lungs, liver, bones, and the orbits may occur and occasionally results in death.
Causes
Granuloma inguinale is caused by Klebsiella granulomatis, a gram-negative pleomorphic bacillus formerly known as Calymmatobacterium granulomatis.
Laboratory Studies
- Although isolation of Klebsiella granulomatis has been reported, the organism is extremely fastidious and culture is beyond the capability of most laboratories. The easiest method to visualize the organism is via smears from the base of the ulcer. The organisms are seen within the cytoplasm of histiocytes. Characteristically, they exhibit bipolar staining, which has been likened to a safety-pin appearance, and are referred to as Donovan bodies. The organisms can occasionally be identified in tissue biopsy specimens with the use of special stains.
- If a quick diagnosis is necessary, a smear can be performed. First, a cotton swab is gently rolled over the ulcer so as not to cause bleeding. The swab is then rolled over a glass slide. The slide is allowed to air dry and is then stained with Giemsa stain or pinacyanol to demonstrate Donovan bodies.
- Alternatively, a crush preparation can be performed. A small piece of tissue should be obtained from the ulcer edge or base via punch biopsy, curettage, or a thin wedge resection. Next, the tissue is crushed between 2 glass slides, separated, and then air dried. A Wright-Giemsa, Warthin-Starry, toluidine blue, or Leishman stain may be used to demonstrate the Donovan bodies.
- Lastly, a tissue biopsy specimen can be obtained; however, the organisms may be difficult to find in early or secondarily infected lesions, or on routine stained sections with hematoxylin and eosin. Thin, paraffin-embedded sections stained with Giemsa or silver stain may facilitate identification of the rod-shaped, encapsulated organisms within the macrophages.
- Polymerase chain reaction techniques may be more sensitive ; however, they are currently only used for scientific research.
- An indirect immunofluorescent technique is available to test serum; however, it is not accurate enough for confirmatory diagnosis.
- Culture of Klebsiella granulomatis from feces has been reported using a monocyte co-culture system and a modified Chlamydia culture.
- Papanicolaou smears may identify Donovan bodies in patients undergoing routine cervical cytological screening.
Imaging Studies
- If bony involvement is suspected in granuloma inguinale, radiography or other imaging studies are indicated.
Other Tests
- Testing for other sexually transmitted diseases is warranted because multiple coexisting infections are common.
Histologic Findings
The epidermis displays acanthosis at the ulcer edge, with pseudoepitheliomatous hyperplasia variably present. A dense dermal infiltrate of histiocytes and plasma cells is present, with a scattering of small neutrophilic abscesses. The macrophages are large and vacuolated, and they contain intracellular bacilli (ie, Donovan bodies), which are best visualized using special stains such as a Warthin-Starry, Wright-Giemsa, or Leishman stain. Klebsiella granulomatis does not stain well with hematoxylin and eosin.
Laboratory Studies
- Although isolation of Klebsiella granulomatis has been reported, the organism is extremely fastidious and culture is beyond the capability of most laboratories. The easiest method to visualize the organism is via smears from the base of the ulcer. The organisms are seen within the cytoplasm of histiocytes. Characteristically, they exhibit bipolar staining, which has been likened to a safety-pin appearance, and are referred to as Donovan bodies. The organisms can occasionally be identified in tissue biopsy specimens with the use of special stains.
- If a quick diagnosis is necessary, a smear can be performed. First, a cotton swab is gently rolled over the ulcer so as not to cause bleeding. The swab is then rolled over a glass slide. The slide is allowed to air dry and is then stained with Giemsa stain or pinacyanol to demonstrate Donovan bodies.
- Alternatively, a crush preparation can be performed. A small piece of tissue should be obtained from the ulcer edge or base via punch biopsy, curettage, or a thin wedge resection. Next, the tissue is crushed between 2 glass slides, separated, and then air dried. A Wright-Giemsa, Warthin-Starry, toluidine blue, or Leishman stain may be used to demonstrate the Donovan bodies.
- Lastly, a tissue biopsy specimen can be obtained; however, the organisms may be difficult to find in early or secondarily infected lesions, or on routine stained sections with hematoxylin and eosin. Thin, paraffin-embedded sections stained with Giemsa or silver stain may facilitate identification of the rod-shaped, encapsulated organisms within the macrophages.
- Polymerase chain reaction techniques may be more sensitive ; however, they are currently only used for scientific research.
- An indirect immunofluorescent technique is available to test serum; however, it is not accurate enough for confirmatory diagnosis.
- Culture of Klebsiella granulomatis from feces has been reported using a monocyte co-culture system and a modified Chlamydia culture.
- Papanicolaou smears may identify Donovan bodies in patients undergoing routine cervical cytological screening.
Imaging Studies
- If bony involvement is suspected in granuloma inguinale, radiography or other imaging studies are indicated.
Other Tests
- Testing for other sexually transmitted diseases is warranted because multiple coexisting infections are common.
Histologic Findings
The epidermis displays acanthosis at the ulcer edge, with pseudoepitheliomatous hyperplasia variably present. A dense dermal infiltrate of histiocytes and plasma cells is present, with a scattering of small neutrophilic abscesses. The macrophages are large and vacuolated, and they contain intracellular bacilli (ie, Donovan bodies), which are best visualized using special stains such as a Warthin-Starry, Wright-Giemsa, or Leishman stain. Klebsiella granulomatis does not stain well with hematoxylin and eosin.
Medical Care
The recommended antibiotic for granuloma inguinale is either trimethoprim/sulfamethoxazole7 or doxycycline. Alternatives include ciprofloxacin, erythromycin, or azithromycin.8 The antibiotic should be given for at least a 3-week course and continued until reepithelialization of the ulcer occurs and any signs of the disease have resolved. If the granuloma inguinale ulcers do not respond within the first days of therapy, add an aminoglycoside (eg, gentamicin at 1 mg/kg IV q8h). Relapse of granuloma inguinale may occur up to 18 months after treatment. In some countries, tetracycline is no longer recommended, owing to bacterial resistance.9
Special considerations
Pregnancy is a relative contraindication for the use of sulfonamides. In pregnant and lactating women with granuloma inguinale, the Centers for Disease Control and Prevention10 recommends erythromycin with or without a parenteral aminoglycoside; however, recent data suggest erythromycin may increase the risk of congenital malformation.11 Doxycycline and ciprofloxacin are contraindicated in pregnancy.
HIV-associated granuloma inguinale may take longer to heal, and the addition of a parenteral aminoglycoside to the regimen is highly recommended.12
Sexual contacts within 60 days prior to the onset of the patient’s symptoms of granuloma inguinale should be examined and offered therapy.
Surgical Care
Once granuloma inguinale is healed, disfiguring genital swellings may need to be surgically corrected.
Medication
The goal of pharmacotherapy for granuloma inguinale is to reduce morbidity and to prevent complications.
Antibiotics
Empiric antimicrobial therapy must be comprehensive and should cover all likely pathogens in the context of the clinical setting.
Trimethoprim/sulfamethoxazole (Bactrim IV, Bactrim SS, Bactrim DS, Septra)
Sulfamethoxazole inhibits bacterial synthesis of dihydrofolic acid. Trimethoprim reversibly inhibits dihydrofolate reductase and blocks the production of tetrahydrofolic acid from dihydrofolic acid.
Adult
One DS tab (800 mg/160 mg) PO bid for at least 3 wk
Pediatric
<2 months: Contraindicated
>2 months: 15-20 mg/kg/d (based on TMP) PO divided q6-8h for at least 3 wk
May increase PT when used with warfarin (perform coagulation tests and adjust dose accordingly); coadministration of diuretics, primarily thiazides, increases incidence of thrombocytopenia purpura in elderly persons; phenytoin levels may increase with coadministration; sulfonamides may increase free methotrexate concentrations; TMP-SMZ may interfere with Jaffe alkaline picrate reaction for creatinine, causing overestimation of creatinine value
Documented hypersensitivity; megaloblastic anemia due to folate deficiency; pregnant patients; nursing mothers; age <2 mo
Pregnancy
C – Fetal risk revealed in studies in animals but not established or not studied in humans; may use if benefits outweigh risk to fetus
Precautions
Discontinue at first appearance of rash or sign of adverse reaction; Stevens-Johnson syndrome, toxic epidermal necrolysis, fulminant hepatic necrosis, agranulocytosis, aplastic anemia, and other blood dyscrasias have rarely occurred; pseudomembranous colitis caused by Clostridium difficile has been reported; sulfonamide hypersensitivity reactions may cause cough, shortness of breath, and pulmonary infiltrates
Give with caution to patients with impaired renal or hepatic function, possible folate deficiency (elderly persons, alcoholic patients, anticonvulsant therapy, malabsorption, malnutrition) and those with severe allergies or bronchial asthma; dose-related hemolysis may occur in patients with G-6-PD deficiency
Obtain CBC count frequently and discontinue therapy if significant hematologic changes occur; may cause bone marrow suppression (if signs occur, give 5-15 mg/d leucovorin); patients with AIDS may not tolerate or respond to TMP-SMZ in same manner as non-AIDS patients; give fluids to prevent crystalluria and stone formation
Give with caution to patients with impaired renal or hepatic function, possible folate deficiency (elderly, alcoholic, anticonvulsant therapy, malabsorption, malnutrition) and those with severe allergies or bronchial asthma; dose-related hemolysis may occur in patients with G-6-PD deficiency
Obtain CBC count frequently and discontinue therapy if significant hematologic changes occur; may cause bone marrow suppression (if signs occur, give 5-15 mg/d leucovorin); patients with AIDS may not tolerate or respond to TMP-SMZ in same manner as non-AIDS patients; give fluids to prevent crystalluria and stone formation
Doxycycline (Adoxa, Doryx, Vibramycin, Periostat)
Bacteriostatic tetracycline antibiotic that inhibits protein synthesis and thus bacterial growth by binding to 30S and possibly 50S ribosomal subunits of susceptible bacteria.
Adult
100 mg PO bid for at least 3 wk
Pediatric
<8 years: Not recommended
>8 years and <45 kg: 2.2 mg/kg PO/IV bid for at least 3 wk; not to exceed 200 mg/d
>8 years and >45 kg: 100 mg PO bid for at least 3 wk
Bioavailability decreases with antacids containing aluminum, calcium, magnesium, iron, or bismuth subsalicylate; depresses plasma prothrombin activity (anticoagulant dosage may need to be decreased); decreases effect of penicillin; barbiturates, carbamazepine, and phenytoin decrease half-life; concurrent use with methoxyflurane has resulted in fatal renal toxicity; concurrent use with oral contraceptives may render them less effective
Documented hypersensitivity
Pregnancy
D – Fetal risk shown in humans; use only if benefits outweigh risk to fetus
Precautions
Photosensitivity may occur with prolonged exposure to sunlight or tanning equipment; caution in patients with hepatic or renal insufficiency; reduce dose in renal impairment; consider drug serum level determinations in prolonged therapy; use during tooth development (last half of pregnancy through age 8 y) can cause permanent discoloration of teeth; Fanconilike syndrome may occur with outdated tetracyclines; as with all antibiotics, risk of superinfection exists; bulging fontanels in infants and benign intracranial hypertension in adults may occur (these resolve upon discontinuation)
Ciprofloxacin (Cipro)
Bactericidal fluoroquinolone antibiotic that inhibits the bacterial enzymes topoisomerase II (DNA gyrase) and topoisomerase IV, which are required for bacterial DNA replication, transcription, repair, and recombination.
Adult
750 mg PO bid for at least 3 wk
Pediatric
<18 years: Not recommended
>18 years: Administer as in adults
Multivalent cation-containing products such as magnesium/aluminum antacids, sucralfate, didanosine, other highly buffered drugs or products containing calcium, iron, or zinc may reduce serum levels; administer antacids 2-4 h before or after taking fluoroquinolones; phenytoin serum levels may be altered (increased or decreased); probenecid may increase serum concentrations; may increase toxicity of theophylline, caffeine, tizanidine, glyburide, methotrexate, cyclosporine, and digoxin (monitor theophylline and digoxin levels); may increase effects of warfarin (monitor PT); metoclopramide accelerates absorption of oral ciprofloxacin; NSAIDs (but not acetyl salicylic acid) with very high doses of fluoroquinolones may provoke convulsions
Documented hypersensitivity; use with tizanidine
Pregnancy
C – Fetal risk revealed in studies in animals but not established or not studied in humans; may use if benefits outweigh risk to fetus
Precautions
In prolonged therapy, perform periodic evaluations of organ system functions (eg, renal, hepatic, hematopoietic); adjust dose in renal function impairment; superinfections with drug-resistant bacteria may occur with prolonged or repeated antibiotic therapy; avoid excessive sunlight due to risk of phototoxicity; crystalluria may occur if urine is alkaline or concentrated; CNS may be affected, resulting in nervousness, agitation, insomnia, anxiety, nightmares, or paranoia
Erythromycin (E-Mycin, Ery-Tab, Eryc)
Macrolide antibiotic that inhibits bacterial protein synthesis by binding to 50S ribosomal subunits of susceptible organisms; may be bacteriocidal or bacteriostatic depending on concentration and type of microorganism.
Adult
500 mg PO qid for at least 3 wk
Pediatric
30-50 mg/kg/d PO divided q6-8h for at least 3 wk
Coadministration may increase toxicity of theophylline and digoxin; may potentiate anticoagulant effects of oral anticoagulants; because erythromycin is an inhibitor of cytochrome P450 3A (CYP3A), the following drug levels may rise, causing increased risk of toxicity: ergotamine, dihydroergotamine, benzodiazepines (eg, triazolam, alprazolam, midazolam), HMG-CoA reductase inhibitors (eg, lovastatin, simvastatin), sildenafil, cyclosporine, carbamazepine, tacrolimus, alfentanil, disopyramide, rifabutin, quinidine, methylprednisolone, cilostazol, vinblastine, and bromocriptine; additional drug interactions exist with hexobarbital, phenytoin, valproate, terfenadine, and astemizole
When coadministered with cisapride, terfenadine, or astemizole, rare cases of serious cardiovascular adverse events, including ECG QT/QTc interval prolongation, cardiac arrest, torsades de pointes, and other ventricular arrhythmias, have been observed and fatalities have been reported
Documented hypersensitivity; coadministration of terfenadine, astemizole, pimozide, or cisapride
Pregnancy
B – Fetal risk not confirmed in studies in humans but has been shown in some studies in animals
Precautions
Caution with impaired hepatic function; estolate formulation may cause cholestatic jaundice; adverse gastrointestinal effects are common; discontinue use if nausea, vomiting, malaise, abdominal colic, or fever occur; risk of superinfection; may aggravate weakness in patients with myasthenia gravis; possible increased risk of infantile hypertrophic pyloric stenosis in infants that have consumed erythromycin
Azithromycin (Zithromax)
Azalide antibiotic (subclass of macrolide antibiotics) that inhibits bacterial protein synthesis by binding 50S ribosomal subunits of susceptible organisms; may be bacteriocidal or bacteriostatic depending on concentration and type of microorganism.
Adult
1 g PO qwk for at least 3 wk
Pediatric
<16 years: Not established
>16 years: Administer as in adults