Diabetic foot
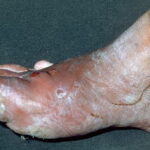
Foot infections are the most common problems in persons with diabetes. These individuals are predisposed to foot infections because of a compromised vascular supply secondary to diabetes. Local trauma and/or pressure (often in association with lack of sensation because of neuropathy), in addition to microvascular disease, may result in various diabetic foot infections. For additional information, see Medscape’s Diabetic Microvascular Complications Resource Center.
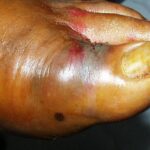
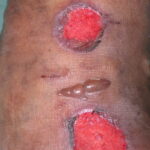
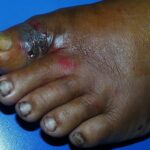
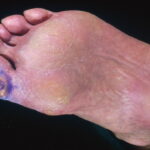
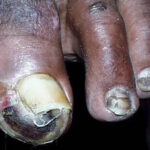
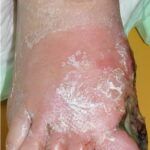
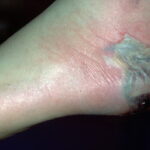
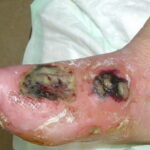
The spectrum of foot infections in diabetes ranges from simple superficial cellulitis to chronic osteomyelitis. Infections in patients with diabetes are difficult to treat because these patients have impaired microvascular circulation, which limits the access of phagocytic cells to the infected area and results in a poor concentration of antibiotics in the infected tissues. For this reason, cellulitis is the most easily treatable and reversible form of foot infections in patients with diabetes. Deep skin and soft tissue infections are also usually curable, but they can be life threatening and result in substantial long-term morbidity.
In terms of the infecting microorganisms and the likelihood of successful treatment with antimicrobial therapy, acute osteomyelitis in people with diabetes is essentially the same as in those without diabetes. Chronic osteomyelitis in patients with diabetes mellitus is the most difficult infection to cure. Adequate surgical debridement, in addition to antimicrobial therapy, is necessary to cure chronic osteomyelitis.
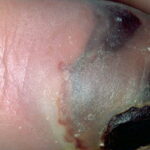
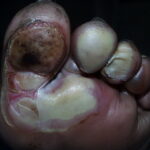
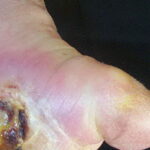
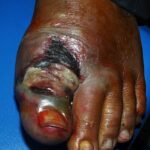
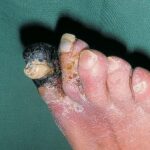
Patients with diabetes also can have a combined infection involving bone and soft tissue called fetid foot. This extensive, chronic soft tissue and bone infection causes a foul exudate and usually requires extensive surgical debridement and/or amputation.
Individuals with diabetes may also have peripheral vascular disease that involves the large vessels, in addition to microvascular and capillary disease that results in peripheral vascular disease with gangrene. Dry gangrene is usually managed with expectant care, and gross infection is usually not present. Wet gangrene usually has an infectious component and requires surgical debridement and/or antimicrobial therapy to control the infection.
Except for chronic osteomyelitis, infections in patients with diabetes are caused by the same microorganisms that can infect the extremities of those without diabetes. Gas gangrene is conspicuous because of its low incidence in patients with diabetes, but deep skin and soft tissue infections, which are due to gas-producing organisms, frequently occur in patients with diabetes. In general, people with diabetes have infections that are more severe and take longer to cure than equivalent infections in other people.
Diabetes mellitus is a disorder that primarily affects the microvascular circulation. In the extremities, microvascular disease due to “sugar-coated capillaries” limits the blood supply to the superficial and deep structures. Pressure due to ill-fitting shoes or trauma further compromises the local blood supply at the microvascular level, predisposing the patient to infection. The infection may involve the skin, soft tissues, bone, or all of these tissues.
Diabetes also accelerates macrovascular disease, which is evident clinically as accelerating atherosclerosis and/or peripheral vascular disease. Most diabetic foot infections occur in the setting of good dorsalis pedis pulses; this finding indicates that the primary problem in diabetic foot infections is microvascular compromise. Impaired microvascular circulation hinders white cell migration into the area of infection and limits the ability of antibiotics to reach the site of infection in an effective concentration. Diabetic neuropathy may be encountered in conjunction with vasculopathy. This may allow for incidental trauma that goes unrecognized (eg, blistering, penetrating foreign body).
In chronic osteomyelitis, a sequestrum and involucrum form; these represent islands of infected bone. Bone fragments that are isolated have no blood supply. Administered antibiotics do not penetrate the devascularized infected bone fragments; they can enter the area of osteomyelitis only via the remaining blood supply. Therefore, antibiotic therapy alone cannot cure patients with chronic osteomyelitis without surgical debridement to remove these isolated infected elements. Surgical debridement is essential to remove the infected bony fragments that the antibiotics cannot reach so that affected areas can be treated with antimicrobial therapy.
Physical
Findings after physical examination may include the following:
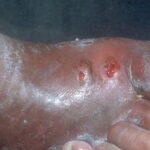
- Cellulitis
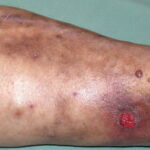
- Cellulitis may involve tender and erythematous nonraised skin lesions on the lower extremity that may or may not be accompanied by lymphangitis.
- Lymphangitis suggests a group A streptococcal etiology.
- If bullae are present, Staphylococcus aureus is the most likely pathogen, but group A streptococci occasionally causes bullous lesions.
- No ulcer or wound exudate is present in patients with cellulitis.
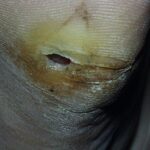
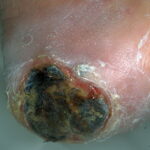
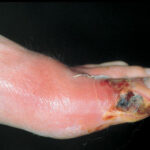
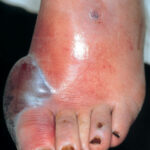
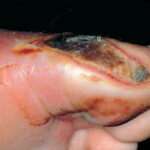
- Deep skin and soft tissue infections
- Patients with deep skin and soft tissue infections may be acutely ill, with painful induration of the soft tissues in the extremity.
- These infections are particularly common in the thigh area, but they may be seen anywhere on the leg or foot.
- Wound discharge is usually not present. In mixed infections that may involve anaerobes, crepitation may be noted over the afflicted area.
- Extreme pain and tenderness indicate the possibility of a compartment syndrome, which may be diagnosed with the aid of a CT scan. Similarly, extreme pain may be an indication of infection with clostridial species (ie, gas gangrene).
- The tissues are not tense, and bullae may be present.
- If a discharge is present, it is often foul.
- Acute osteomyelitis
- Unless peripheral neuropathy is present, the patient has pain at the site of the involved bone.
- Usually, fever and regional adenopathy are absent.
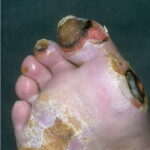
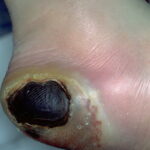
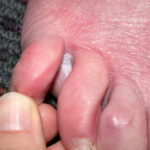
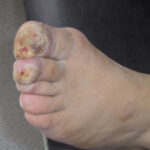
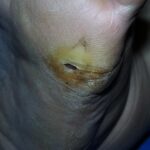
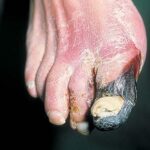
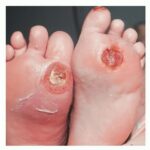
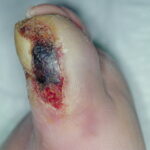
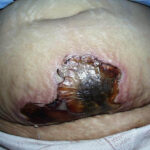
- Chronic osteomyelitis
- In chronic osteomyelitis, the patient’s temperature is usually less than 102°F.
- Discharge is commonly foul.
- No lymphangitis is observed.
- Pain may or may not be present, depending on the degree of peripheral neuropathy.
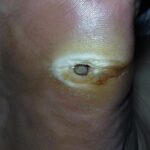
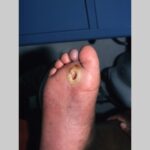
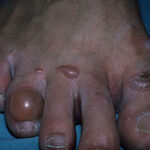
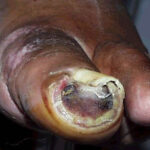
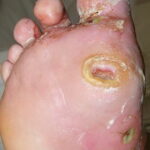
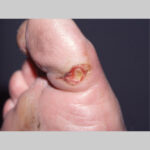
- The deep penetrating ulcers and sinuses are usually located between the toes or on the plantar surface of the foot.
- In patients with diabetes, chronic osteomyelitis usually does not occur on the medial malleoli, shins, or heels.
- Importantly, deep penetrating foot ulcers or deep sinus tracts are diagnostic of chronic osteomyelitis.
Causes
The microbiologic features of diabetic foot infections vary according to the tissue infected.
- In patients with diabetes, superficial skin infections such as cellulitis are caused by the same organisms as those in healthy hosts, namely group A streptococci and S aureus. However, in unusual epidemiologic circumstances, organisms such as Pasteurella multocida (eg, from dog or cat bites or scratches) may be noted and should always be considered.
- Group B streptococcal cellulitis is uncommon in healthy hosts and not uncommon in patients with diabetes. In people with diabetes, group B streptococci may cause urinary tract infections and catheter-associated bacteriuria in addition to cellulitis, skin and/or soft tissue infections, and chronic osteomyelitis. Such infections may be complicated by bacteremia.
- In patients with diabetes, deep soft tissue infections can be associated with gas-producing gram-negative bacilli. Clinically, these infections appear as necrotizing fasciitis, compartment syndrome, or myositis. Gas gangrene is uncommon in persons with diabetes.
- Acute osteomyelitis usually occurs as a result of foot trauma in an individual with diabetes. The distribution of organisms is the same as in an individual without diabetes who has acute osteomyelitis.
- In chronic osteomyelitis, the pathogens are group A and group B streptococci, aerobic gram-negative bacilli, and Bacteroides fragilis, among others.
- Pseudomonas aeruginosa is generally not a pathogen in chronic osteomyelitis in patients with diabetes.
- P aeruginosa is frequently cultured from samples obtained from a draining sinus tract or deep penetrating ulcers in patients with diabetes. However, these organisms are superficial colonizers and are generally not the cause of the bone infection.
- Because Pseudomonas organisms are water-borne, superficial ulcers may be contaminated by bacteria in wet socks or dressings. To the author’s knowledge, no well-documented cases of biopsy-proven P aeruginosa infection have been reported in patients with chronic osteomyelitis.
- Bone biopsy performed under aseptic conditions in the operating room reveals that chronic osteomyelitis in patients with diabetes is not due to P aeruginosa.
- B fragilis is an important bone pathogen in chronic osteomyelitis in patients with diabetes.
- Other pathogens implicated in chronic osteomyelitis in patients with diabetes include Escherichia coli, Proteus mirabilis, and Klebsiella pneumoniae.
- Fetid foot represents a combined deep skin and soft tissue infection caused by pathogens involved in chronic osteomyelitis.
Laboratory Studies
Cellulitis
- The CBC count and erythrocyte sedimentation rate (ESR) are slightly or moderately elevated in cellulitis. Elevations are not diagnostic and, therefore, are unhelpful.
- Blood culture results are usually negative. If positive, they usually indicate the presence of group A or group B streptococci. Cultures of skin via aspiration or biopsy are generally unrewarding.
Skin and soft tissue infections
- The CBC count and ESR are mildly or moderately elevated.
- If bullae are present, Gram stain and culture results in aspirated exudate from a bullous lesion may provide clues to the etiology of the infection.
- Blood culture results may be positive.
Acute osteomyelitis
- The CBC count usually reveals leukocytosis, and the ESR is moderately or highly elevated.1
- Blood culture results are usually negative. When positive, the findings most frequently indicate the presence of S aureus.
Chronic osteomyelitis
- The CBC count is often within the reference range. Usually, the ESR is very highly elevated; it may exceed 100 mm/h.1
- The platelet count is often elevated in chronic osteomyelitis.
- Blood culture results are usually negative in patients with chronic osteomyelitis.
Imaging Studies
Cellulitis: Image studies are not applicable.
Deep skin and soft tissue infections
- In a patient with diabetes considered to have a deep soft tissue infection, plain radiography, CT scan, or MRI may be performed to rule out a compartment syndrome and to demonstrate the presence of gas or a foreign body in the deep tissues.
- A finding of excessive gas signifies a mixed aerobic-anaerobic infection in contrast to gas gangrene (clostridial myonecrosis).
Acute osteomyelitis
- For long bones, plain radiographic findings generally become abnormal after 10-14 days. Soft tissue swelling and periosteal elevation are the earliest signs of acute osteomyelitis on a plain radiograph.
- Bone scan findings are positive within 24 hours in acute osteomyelitis.
- A bone scan is preferred to gallium or indium scans in the assessment of acute osteomyelitis.
- Gallium or indium scans offer no additional information, and the findings are not more specific than those of bone scans in the diagnosis of osteomyelitis.
- Indium scans often show false-negative results in acute or chronic osteomyelitis.
Chronic osteomyelitis
- When osteomyelitis becomes chronic, plain radiographic findings are invariably abnormal.
- Bone scans are usually unnecessary unless diagnostic confusion exists with another disorder.
- A bone tumor is best differentiated from chronic osteomyelitis with the aid of bone scanning or MRI prior to definitive bone biopsy.
Other Tests
Patients with diabetic foot infections and peripheral vascular disease may benefit from vascular surgical evaluation to bypass large-vessel occlusive disease. However, large-vessel bypass does not cure the microvascular component of diabetic foot infections.
Procedures
Aspiration of a sample from the leading edge of the erythematous border in a patient with cellulitis is usually not necessary, but a sample may be aspirated if the likely organism must be identified on initial presentation. However, the yield is low, likely to be less than 5%.
Samples from deep skin and soft tissue infections may be aspirated. Gram stains and/or cultures may be used to identify the organism.
Aside from blood culturing, radiography, and nuclear imaging studies, bone biopsy is not necessary in acute osteomyelitis because the pathogens are predictable.
Bone biopsy performed under aseptic conditions in the operating room is the preferred way to identify the causative pathogen in chronic osteomyelitis. Because surgical debridement is critical in treating chronic osteomyelitis, bone biopsy specimens are usually not obtained during the surgical debridement procedure.
Histologic Findings
Involucrum and/or sequestrum may be present in the cortical bone in cases of chronic osteomyelitis. Subperiosteal elevation and/or infection may involve the cortex in acute osteomyelitis.
Staging
Staging is applicable only in cases of chronic osteomyelitis that require surgery
Medical Care
Immobilization is important in acute or chronic osteomyelitis.
Surgical Care
Chronic osteomyelitis cannot be cured without adequate surgical debridement.
Consultations
Appropriate consultation with a surgeon should be obtained for debridement and/or amputation in chronic osteomyelitis.
A general or vascular surgeon should be consulted for the debridement or decompression of compartment syndromes in patients with deep skin and soft tissue infections.
An infectious disease specialist should be consulted in the treatment of all patients with diabetic foot infections to optimize the antimicrobial therapy.
Diet
Dietary modifications are not applicable except to optimize diabetes control.
Activity
The patient may participate in activities as tolerated. However, weight-bearing may be contraindicated.
Medication
In patients with diabetes, cellulitis is generally caused by group A and group B streptococci and, occasionally, S aureus. Cellulitis may be treated with single antibiotics that have the appropriate spectrum or with combination therapy that covers the appropriate organisms.
Antibiotics
Appropriate monotherapy for cellulitis includes cefazolin or clindamycin. Although gram-negative organisms are the unusual causes of cellulitis, even in diabetes, if they are suspected, a fluoroquinolone (eg, levofloxacin) may be used in conjunction with clindamycin.
In patients with diabetes, deep skin and severe soft tissue infections are usually due to mixed aerobic and anaerobic organisms. These infections may be treated with monotherapy involving meropenem or piperacillin and tazobactam. Alternatively, clindamycin plus levofloxacin or metronidazole may be used.
Acute osteomyelitis, which usually is due to S aureus, may be treated with cefazolin, clindamycin, and an antistaphylococcal penicillin (eg, nafcillin).
In chronic osteomyelitis, coverage must be directed against S aureus, group A and group B streptococci, aerobic gram-negative bacilli (excluding P aeruginosa), and B fragilis. Monotherapy for chronic osteomyelitis may include ampicillin and sulbactam, piperacillin and tazobactam, or meropenem. In chronic osteomyelitis, antimicrobial therapy without adequate debridement does not eliminate the infection.
Combination therapy for diabetic foot infections involves levofloxacin plus clindamycin.
Meropenem (Merrem)
Bactericidal broad-spectrum carbapenem antibiotic that inhibits cell-wall synthesis. Effective against most gram-positive and gram-negative bacteria. Has slightly increased activity against gram-negative species and slightly decreased activity against staphylococci and streptococci compared with imipenem. Structurally similar to beta-lactam antibiotics, but not related to beta-lactam drugs in terms of hypersensitivity. Carbapenems and monobactams do not cross-react in patients who are allergic to penicillin, even those who have an anaphylactic reaction to penicillin.
Adult
1 g IV q8h
Pediatric
40 mg/kg IV q8h
Probenecid may inhibit renal excretion, increasing levels
Documented hypersensitivity
Pregnancy
B – Fetal risk not confirmed in studies in humans but has been shown in some studies in animals
Precautions
Pseudomembranous colitis and thrombocytopenia (immediately discontinue)
Ampicillin and sulbactam (Unasyn)
Drug combination of a beta-lactamase inhibitor with ampicillin. Covers skin, enteric flora, and anaerobes. Not ideal for nosocomial pathogens.
Adult
1.5-3 g (1 g ampicillin and 0.5 g sulbactam to 2 g ampicillin and 1 g sulbactam) IV q6h; not to exceed 4 g/d sulbactam or 8 g/d ampicillin
Pediatric
<3 months: Not established
3 months to 12 years: 100-200 mg ampicillin/kg/d (150-300 mg Unasyn) IV divided q6h
>12 years: Administer as in adults
Probenecid and disulfiram elevate ampicillin levels; allopurinol decreases ampicillin effects and has additive effect on ampicillin rash; may decrease effect of oral contraceptives
Documented hypersensitivity
Pregnancy
B – Fetal risk not confirmed in studies in humans but has been shown in some studies in animals
Precautions
Adjust dose in renal failure; evaluate rash and differentiate from hypersensitivity reaction
Piperacillin and tazobactam (Zosyn)
Antipseudomonal penicillin plus a beta-lactamase inhibitor. Inhibits biosynthesis of cell wall mucopeptide and is effective during the active multiplication stage.
Adult
4 g IV q8h
Pediatric
75 mg/kg IV q6h
Tetracyclines may decrease piperacillin effects; high concentrations of piperacillin may physically inactivate aminoglycosides if administered in the same IV line; synergistic effects with concurrent aminoglycosides; probenecid may increase penicillin levels; high-dose parenteral penicillins may increase risk of bleeding
Documented hypersensitivity; do not use oral penicillin in severe pneumonia, bacteremia, pericarditis, emphysema, meningitis, and purulent or septic arthritis during the acute stage
Pregnancy
B – Fetal risk not confirmed in studies in humans but has been shown in some studies in animals
Precautions
Assess CBC count before therapy and at least weekly during therapy; during therapy, monitor AST and ALT for liver function abnormality; caution in hepatic insufficiency; perform urinalysis and determine BUN and creatinine levels during therapy (adjust dose if elevated); monitor blood levels (possible neurotoxic reaction)
Cefazolin (Kefzol, Ancef, Zolicef)
First-generation semisynthetic cephalosporin that arrests bacterial cell-wall synthesis, inhibiting bacterial growth. Primarily active against skin flora, including S aureus.
Typically used alone for skin and skin-structure coverage. IV/IM dosing regimens are similar
Adult
1 g IV q8h
Pediatric
25-100 mg/kg/d IV/IM divided q6-8h depending on severity of infection; not to exceed 6 g/d
Probenecid prolongs effect; coadministration with aminoglycosides may increase renal toxicity; may yield false-positive urine-dip results for glucose
Documented hypersensitivity
Pregnancy
B – Fetal risk not confirmed in studies in humans but has been shown in some studies in animals
Precautions
Adjust dose in severe renal insufficiency (high doses may cause CNS toxicity); superinfections and promotion of nonsusceptible organisms may occur with prolonged or repeated therapy
Clindamycin (Cleocin)
Lincosamide for the treatment of serious skin and soft tissue staphylococcal infections.
Also effective against aerobic and anaerobic streptococci, except enterococci. Inhibits bacterial growth, possibly by blocking dissociation of peptidyl t-RNA from ribosomes, arresting RNA-dependent protein synthesis.
Adult
150-300 mg/dose PO q8h; not to exceed 1.8 g/d
Up to 900 mg IV q8h depending on degree of infection
Pediatric
8-20 mg/kg/d PO as hydrochloride or 8-25 mg/kg/d as palmitate divided tid/qid
20-40 mg/kg/d IV/IM divided tid/qid
Increases duration of neuromuscular blockade induced by tubocurarine and pancuronium; erythromycin may antagonize effects; antidiarrheals may delay absorption
Documented hypersensitivity; regional enteritis, ulcerative colitis, hepatic impairment, and antibiotic-associated colitis
Pregnancy
B – Fetal risk not confirmed in studies in humans but has been shown in some studies in animals
Precautions
Adjust dose in severe hepatic dysfunction; no adjustment necessary in renal insufficiency; associated with severe and possibly fatal colitis by allowing overgrowth of Clostridium difficile
Levofloxacin (Levaquin)
Used to treat complicated and uncomplicated skin and skin structure infections. Fluoroquinolones should be used empirically in patients likely to develop exacerbation due to resistant organisms to other antibiotics. This is the L stereoisomer of the D/L parent compound ofloxacin, the D form being inactive. Good monotherapy with extended coverage against Pseudomonas species, as well as excellent activity against pneumococci. Agent acts by inhibition of DNA gyrase activity. PO form has bioavailability that reportedly is 99%.
Adult
500 mg PO/IV q24h
Pediatric
<18 years: Not recommended
>18 years: Administer as in adults
Antacids, iron salts, and zinc salts may reduce serum levels; administer antacids 2-4 h before or after fluoroquinolone use; cimetidine may interfere with metabolism of fluoroquinolones; reduces therapeutic effects of phenytoin; probenecid may increase serum concentrations; may increase toxicity of theophylline, caffeine, cyclosporine, and digoxin (monitor digoxin levels); may increase effects of anticoagulants (monitor PT)
Documented hypersensitivity
Pregnancy
C – Fetal risk revealed in studies in animals but not established or not studied in humans; may use if benefits outweigh risk to fetus
Precautions
Periodically evaluate organ (eg, renal, hepatic, hematopoietic) function in prolonged therapy; adjust dose in renal impairment; superinfections may occur with prolonged or repeated antibiotic therapy
Metronidazole (Flagyl, Protostat)
Imidazole ring-based antibiotic active against various anaerobic bacteria and protozoa. Used in combination with other antimicrobial agents. Not used for C difficile enterocolitis.
Adult
1 g IV q24h or 250-500 mg PO q6-8h
Pediatric
Administer as in adults
May increase toxicity of anticoagulants, lithium, and phenytoin; cimetidine may increase toxicity; disulfiram reaction possible with alcohol ingestion
Documented hypersensitivity
Pregnancy
B – Fetal risk not confirmed in studies in humans but has been shown in some studies in animals
Precautions
Adjust dose in hepatic disease; monitor for seizures and peripheral neuropathy
Nafcillin (Unipen, Nafcil, Nallpen)
Initial therapy for suspected penicillin G–resistant streptococcal or staphylococcal infections. Initially, use parenteral therapy in severe infections; change to PO therapy as condition warrants. Because of thrombophlebitis, particularly in elderly patients, use parenteral administration only for a short term (1-2 d); change to PO route as clinically indicated.
Adult
1-2 g IV q4h
Pediatric
<4 kg (neonates): 10 mg/kg IM bid
4-40 kg: 25 mg/kg IM bid, 100-200 mg/kg/d IV/IM in 4-6 divided doses, or 50 mg/kg/d PO divided qid
>40 kg: Administer as in adults
Associated with warfarin resistance when administered concurrently; effects may decrease with bacteriostatic action of tetracycline derivatives
Documented hypersensitivity
Pregnancy
B – Fetal risk not confirmed in studies in humans but has been shown in some studies in animals
Precautions
To optimize therapy, determine causative organisms and susceptibility; treatment is >10 d to eliminate infection and prevent sequelae (eg, endocarditis, rheumatic fever); obtain cultures after treatment to confirm that infection is eradicated
Moxifloxacin (Avelox)
Inhibits A subunits of DNA gyrase, inhibiting bacterial DNA replication and transcription.
Adult
400 mg IV/PO qd
Pediatric
<18 years: Not recommended
>18 years: Administer as in adults
Antacids and electrolyte supplements reduce absorption; loop diuretics, probenecid, and cimetidine increase serum levels; NSAIDs enhance CNS-stimulating effect; may increase toxicity of theophylline, caffeine, cyclosporine, and digoxin (monitor digoxin levels); may increase effects of anticoagulants (monitor PT)
Documented hypersensitivity; Q-T prolongation or concurrent administration of drugs that cause Q-T prolongation
Pregnancy
C – Fetal risk revealed in studies in animals but not established or not studied in humans; may use if benefits outweigh risk to fetus
Precautions
Periodically evaluate organ (eg, renal, hepatic, hematopoietic) function in prolonged therapy; adjust dose in renal impairment; superinfections may occur with prolonged or repeated antibiotic therapy; may induce seizures in CNS disorder
Tigecycline (Tygacil)
A glycylcycline antibiotic that is structurally similar to tetracycline antibiotics. Inhibits bacterial protein translation by binding to 30S ribosomal subunit and blocks entry of amino-acyl tRNA molecules in ribosome A site. Indicated for complicated skin and skin structure infections caused by E coli, E faecalis (vancomycin-susceptible isolates only), S aureus (methicillin-susceptible and -resistant isolates), S agalactiae, S anginosus grp (includes S anginosus, S intermedius, S constellatus), S pyogenes, and B fragilis.
Adult
Infuse each dose over 30-60 min
100 mg IV once, then 50 mg IV q12h
Severe hepatic impairment (ie, Child Pugh class C): 100 mg IV once, then 25 mg IV q12h
Pediatric
<18 years: Not established
>18 years: Administer as in adults
Coadministration decreases warfarin clearance and increases warfarin Cmax and AUC (monitor aPTT and INR); coadministration of antibiotics with oral contraceptives may decrease contraceptive effect
Documented hypersensitivity
Pregnancy
D – Fetal risk shown in humans; use only if benefits outweigh risk to fetus
Precautions
Caution in severe hepatic impairment (reduce dose); may adversely effect tooth development; may permit clostridia overgrowth, resulting in antibiotic-associated colitis; may have adverse effects similar to tetracyclines (eg, photosensitivity, pseudotumor cerebri, pancreatitis, antianabolic action)
Ertapenem (Invanz)
Bactericidal activity results from inhibition of cell wall synthesis and is mediated through ertapenem binding to penicillin binding proteins. Stable against hydrolysis by various beta-lactamases, including penicillinases, cephalosporinases, and extended spectrum beta-lactamases. Hydrolyzed by metallo-beta-lactamases.
Adult
1 g qd for 14 d if IV and 7 d if IM; infuse over 30 min if IV
CrCl 30 mL/min/1.73 m2: 500 mg IV qd
Pediatric
<3 months: Not established
3 months to 12 years: 15 mg/kg IV q12h; not to exceed 1 g/d
>12 years: Administer as in adults
Probenecid may reduce renal clearance of ertapenem and increase half-life but benefit is minimum and does not justify coadministration
Documented hypersensitivity to drug or amide type anesthetics
Pregnancy
B – Fetal risk not confirmed in studies in humans but has been shown in some studies in animals
Precautions
Pseudomembranous colitis may occur; seizures and CNS adverse reactions may occur; when using with lidocaine to administer intramuscularly, avoid inadvertent injection into blood vessel; decrease dose in renal failure; serious and occasionally fatal hypersensitivity reactions may occur with beta lactams, caution with previous hypersensitivity reactions to penicillin, cephalosporins, other beta lactams, or other allergens; do not mix or coinfuse in same IV line as other medications; do not mix with dextrose-containing diluents
Minocycline (Dynacin, Minocin)
Treats infections caused by susceptible gram-negative and gram-positive organisms, in addition to infections caused by susceptible Chlamydia, Rickettsia, and Mycoplasma.
Adult
100 mg PO bid for 5-7 d
Pediatric
<8 years: Not recommended
>8 years: 4 mg/kg PO initially, followed with 2 mg/kg q12h
Bioavailability decreases with antacids containing aluminum, calcium, magnesium, iron, or bismuth subsalicylate; can decrease effects of oral contraceptives, causing breakthrough bleeding and increased risk of pregnancy; tetracyclines can increase hypoprothrombinemic effects of anticoagulants
Documented hypersensitivity; severe hepatic dysfunction
Pregnancy
D – Fetal risk shown in humans; use only if benefits outweigh risk to fetus
Precautions
Photosensitivity may occur with prolonged exposure to sunlight or tanning equipment; reduce dose in renal impairment; consider drug serum level determinations in prolonged therapy; tetracycline use during tooth development (last one-half of pregnancy through age 8 y) can cause permanent discoloration of teeth; Fanconilike syndrome may occur with outdated tetracyclines; hepatitis or lupuslike syndromes may occur
Daptomycin (Cubicin)
First of new antibiotic class called cyclic lipopeptides. Binds to bacterial membranes and causes rapid membrane potential depolarization, thereby inhibiting protein, DNA, and RNA synthesis, and ultimately causing cell death.
Indicated to treat complicated skin and skin structure infections caused by S aureus (including methicillin-resistant strains), S pyogenes, S agalactiae, S dysgalactiae, and E faecalis (vancomycin-susceptible strains only).
Adult
CrCl >30 mL/min: 4 mg/kg IV q24h infused over 30 min
CrCl <30 mL/min: 4 mg/kg IV q48h (including hemodialysis or CAPD)
Pediatric
<18 years: Not established
>18 years: Administer as in adults