Dermatitis artefacta
The skin and nervous system develop adjacent to each other as the ectoderm and neuroectoderm, respectively, in the embryo and remain interconnected throughout life. Dermatitis artefacta is defined as the deliberate and conscious production of self-inflicted skin lesions to satisfy an unconscious psychological or emotional need. These skin lesions serve as powerful, self-expressive, nonverbal messages. Patients often deny responsibility for their creation. Neurotic excoriation is differentiated from dermatitis artefacta by its conscious compulsive nature. Dermatitis artefacta falls under the general category of factitious disorders, which excludes neurotic excoriations, delusional disorders, malingering, and Münchhausen syndrome (except Münchhausen syndrome by proxy).1
Psychiatric conditions, in particular depression,2,3 anxiety,3 personality disorders,3 delusional disorders, and dissociative disorders,4 are often coexistent in 25-33% of all dermatological conditions.5 Dermatitis artefacta may occur in persons of any age and commonly manifests within the context of chronic medical and/or dermatological conditions. These self-induced skin lesions may be present continuously, or they may be episodic, occurring during periods of heightened psychosocial stress and/or uncontrolled psychoses. Patients with dermatitis artefacta require both dermatological assessment and psychosocial support.
Pathophysiology
The pathophysiology of dermatitis artefacta is poorly understood. Multifactorial causes include genetics, psychosocial factors, and personal or family history of psychiatric illness. Commonly, a family member is involved in the medical field, and patients tend to be well versed in medical terminology.
Acute episodes of dermatitis artefacta often represent a maladaptive response to a psychosocial stressor. Long-standing cases may be secondary to underlying anxiety or depression, emotional deprivation, an unstable body image, or a personality disorder with borderline features. Many dermatitis artefacta patients also have an associated chronic medical or dermatological condition.
History
- Dermatitis artefacta patients are usually otherwise healthy and usually do not provide a substantial indicative history, perhaps revealing vague accounts of antecedent events.
- Dermatitis artefacta patients may report a personal history of chronic dermatoses, including acne, alopecia (ie, alopecia areata, androgenic alopecia), atopic dermatitis, chronic idiopathic urticaria, psoriasis, rosacea, or vitiligo.
- They may have a personal or family history of psychiatric illness, including anxiety, depression, personality disorder (ie borderline, dependent, obsessive-compulsive), dissociative disorder, body dysmorphic disorder, or posttraumatic stress disorder. Patients typically are also reluctant to seek a consultation with a psychiatrist.
- They may have a personal history of chronic medical conditions, chronic pain syndromes, or both.
- Dermatitis artefacta patients may report a history of childhood neglect or abuse, sexual abuse, or psychological trauma.
- Münchhausen syndrome by proxy (form of dermatitis artefacta) manifests as skin lesions caused by a parent or caregiver (often the mother). It usually occurs with children younger than 5 years.
- Pertinent information from the history includes the patient’s quality of life with regard to health-related issues, his or her perception of the skin condition, the role of a psychosocial stressor, the presence of a lengthy medical file with numerous consultations, an extensive list of previously used medications, and a history of substance abuse. Also inquire about family members or significant others and their reaction to the skin lesions. Anger, frustration, and impatience are commonly reported reactions.
Physical
Dermatitis artefacta is a challenging clinical diagnosis. It is suggested based on findings that include an absence of other dermatoses to explain the lesions and histological findings that are inconsistent with the clinical presentation.
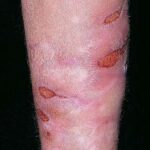
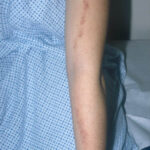
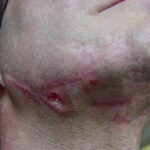
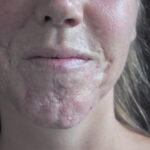
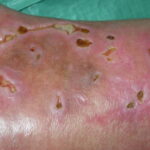
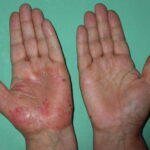
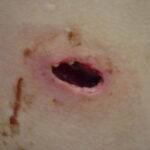
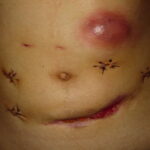
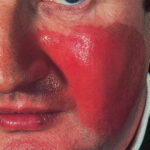
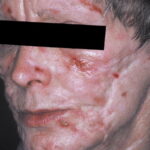
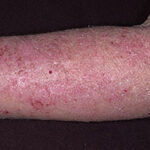
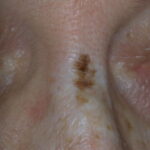
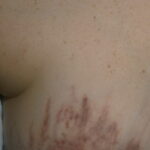
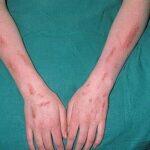
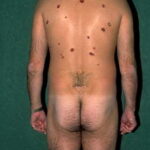
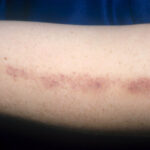
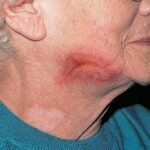
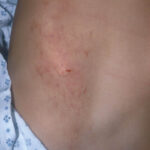
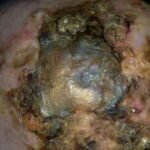
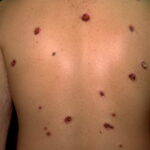
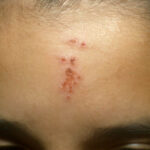
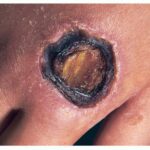
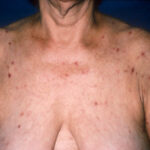
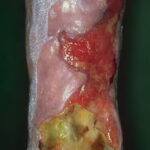
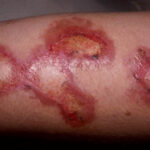
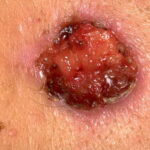
- The morphology of the dermatitis artefacta skin lesions is variable and bizarre and is typically dependent on the mechanism of injury. Features may include sharp margins adjacent to normal skin, geometric shapes, and linear tracks (ie, secondary to exposure to corrosive liquids).
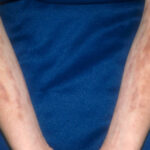
- Most often, the dermatitis artefacta lesions are found at sites accessible to the patient. Typical locations include the face (45%), upper extremity (ie, hand and forearm; 24%), lower extremities (31%), trunk (24%), upper arm (7%), and scalp (7%). Lesions are usually within reach of the dominant hand.
- The appearance of the dermatitis artefacta lesions, in decreasing order of frequency, is as follows:
- Superficial erosion (50%)
- Hyperpigmented macule or purpura (30-42%)
- Excoriation (17%)
- Deep necrosis, ulceration (17%)
- Irritant dermatoses (17%)
- Papules (17%)
- Crusts (8%)
- Scars – Pinpoint, star-shaped, atypically shaped (8%)
- Onychodystrophy
- Other – Keratosis, tattoolike
- Usually, 72% of patients have one type of lesion morphology, 41% have 2 types of lesion morphology, and 31% have 3 types of lesion morphology.
- In 66% of patients, involvement is limited to one body segment. In 34%, involvement is with 2 body segments.
- Dermatitis artefacta must be distinguished from dermatitis neglecta, neurotic excoriations, trichotillomania, and painful bruise syndrome.
- Reported mimics and associations are as follows:
- Pyoderma gangrenosum7,8
- Acquired hemophilia9
- T-cell lymphoma10
- Hepatitis C virus–associated porphyria cutanea tarda.11
Causes
The cause of dermatitis artefacta is multifactorial. One should be sure to rule out hypochondriasis, substance abuse disorder, and psychotic disorders.
- Genetic predisposition: Many psychiatric disorders tend to be familial.
- Psychiatric illness: Associations include anxiety disorders, major depressive disorder, dysthymia, body dysmorphic disorder, factitious disorders, and somatoform disorders.
- Neuropsychological trauma – Child abuse/neglect, sexual abuse, posttraumatic stress disorder
- Personality disorders – Attention-seeking traits (eg, borderline, dependent), obsessions, compulsions
- Psychosocial factors – Poor coping mechanisms, family dysfunction, inadequate social support structure12
- Chronic illnesses: These are commonly associated with dermatitis artefacta.
- Dermatological disorders – Specifically, acne, alopecia (ie, alopecia areata, androgenic alopecia), atopic dermatitis, chronic idiopathic urticaria, psoriasis, rosacea, and vitiligo
- Any long-standing medical illness
- Chronic pain syndromes
Medical Care
Dermatitis artefacta is a challenging condition that requires dermatologic and, often, psychiatric expertise.
- General dermatological care measures include baths, debridement, emollients, and topical antimicrobials.
- A detailed assessment of the patient history for chronic dermatoses, chronic medical conditions, psychiatric illnesses, and psychosocial problems is necessary in the care of dermatitis artefacta.
- An effective therapeutic relationship in dermatitis artefacta patients requires a nonjudgmental, empathetic, and supportive environment. Avoid etiology issues and confrontation. Developing a good rapport with the patient and encouraging the patient to return for follow-up appointments are important.
- A psychiatric evaluation is warranted for severe self-mutilation and any evidence of psychiatric illness, psychosis, risk of suicide, and/or need for hospitalization in dermatitis artefacta patients.
- Complementary adjuvant therapies in dermatitis artefacta patients may include acupuncture, cognitive behavioral therapy (ie, aversion therapy, systemic desensitization, operant conditioning), biofeedback and relaxation therapy (ie, anxiety-related dermatitis artefacta), and hypnosis.13
Medication
Topical antimicrobials are the most commonly prescribed medication; however, topical agents alone have shown limited efficacy. In many instances, treating the underlying psychiatric disorder with antidepressants, antianxiety drugs, and antipsychotic agents is necessary.14,15 Analgesics should be avoided because of the high probability for dependence and addiction.
Topical antimicrobials
Self-inflicted lesions are often accompanied by a localized skin infection.
Bacitracin, Neomycin and Polymyxin B (AK-Spore Ointment, Neocin, Neosporin)
Bacitracin prevents transfer of mucopeptides into growing cell wall, inhibiting bacterial growth.
Neomycin is used for treatment of minor infections; inhibits bacterial protein synthesis and growth. Polymyxin B disrupts bacterial cytoplasmic membrane, permitting leakage of intracellular constituents and causing inhibition of bacterial growth.Adult
Apply 1-4 times/d to affected areas and cover with sterile bandages prn
Pediatric
Administer as in adults
None reported
Documented hypersensitivity; epithelial herpes simplex keratitis; mycobacterial and fungal infections
Pregnancy
C – Fetal risk revealed in studies in animals but not established or not studied in humans; may use if benefits outweigh risk to fetus
Precautions
Caution when treating extensive burns (>20% BSA) because absorption of neomycin is possible and may cause nephrotoxicity and ototoxicity; prolonged use may result in overgrowth of nonsusceptible organisms
Fusidic acid (Fucidin, Fusidin, Fusidin Leo)
Topical antibacterial that inhibits bacterial protein synthesis, causing bacterial death.
Adult
Apply to affected area bid for 2 wk
Pediatric
Administer as in adults
None reported
Documented hypersensitivity
Pregnancy
C – Fetal risk revealed in studies in animals but not established or not studied in humans; may use if benefits outweigh risk to fetus
Precautions
Discontinue if irritation or sensitivity occurs
Oral antibiotics
For impetiginized skin lesions.
Cephalexin (Keflex, Keftab, Biocef)
First-generation cephalosporin that inhibits bacterial replication by inhibiting bacterial cell wall synthesis. Bactericidal and effective against rapidly growing organisms forming cell walls. Resistance occurs by alteration of penicillin-binding proteins. Effective for treatment of infections caused by streptococci or staphylococci, including penicillinase-producing staphylococci. May use to initiate therapy when streptococcal or staphylococcal infection is suspected. Used orally when outpatient management is indicated. Recommended for impetigo caused by Staphylococcus aureus resistant to erythromycin. Primary activity against skin florae. Used for skin infections or prophylaxis in minor procedures.
Adult
500 mg PO q6h for 10 d
Pediatric
25-50 mg/kg/d PO q6h for 10 d; not to exceed 3 g/d
Coadministration with aminoglycosides increases nephrotoxic potential
Documented hypersensitivity
Pregnancy
B – Fetal risk not confirmed in studies in humans but has been shown in some studies in animals
Precautions
Adjust dose in severe renal insufficiency (high doses may cause CNS toxicity); superinfections and promotion of nonsusceptible organisms may occur with prolonged use or repeated therapy
Erythromycin (E-Mycin, Eryc, Ery-Tab)
Use with hypersensitivity or contraindication to penicillin or cephalexin. May result in GI upset, prompting prescription of an alternative macrolide or a change to tid dosing. Covers most potential etiologic agents, including Mycoplasma species.
Less active against Haemophilus influenzae. Although 10 d seems to be a standard course of treatment, treating until patient has been afebrile for 3-5 d seems more rational. Inhibits bacterial growth, possibly by blocking dissociation of peptidyl tRNA from ribosomes, causing RNA-dependent protein synthesis to arrest. For treatment of staphylococcal and streptococcal infections.
In children, age, weight, and severity of infection determine proper dosage. When bid dosing is desired, half the total daily dose may be taken q12h. For more severe infections, double the dose. Has added advantage of being a good anti-inflammatory agent by inhibiting migration of polymorphonuclear leukocytes.Adult
250 mg erythromycin stearate/base (or 400 mg ethylsuccinate) PO q6h, or 500 mg q12h (1 h ac or 2 h pc)
Alternatively, 333 mg PO q8h; increase to 4 g/d depending on severity of infectionPediatric
30-50 mg/kg/d (base or ethylsuccinate) PO divided q6-8h; not to exceed 2 g/d (base) or 3.2 g/d (ethylsuccinate)
Coadministration may increase toxicity of theophylline, digoxin, carbamazepine, and cyclosporine; may potentiate anticoagulant effects of warfarin; coadministration with lovastatin and simvastatin increases risk of rhabdomyolysis; decreases metabolism of repaglinide, thus increasing serum levels and effects
Documented hypersensitivity; hepatic impairment
Pregnancy
B – Fetal risk not confirmed in studies in humans but has been shown in some studies in animals
Precautions
Caution in liver disease; estolate formulation may cause cholestatic jaundice; adverse GI effects are common (give doses pc); discontinue use if nausea, vomiting, malaise, abdominal colic, or fever occurs
Selective serotonin reuptake inhibitors
First-line therapy for depression. Other medications in this category include paroxetine, citalopram, and sertraline. For dermatitis artefacta associated with obsessive-compulsive disorder, the use of an SSRI for at least 6 months to 1 year accompanied by psychotherapy is recommended.
Fluoxetine (Prozac)
Selectively inhibits presynaptic serotonin reuptake with minimal or no effect in reuptake of norepinephrine or dopamine.
May cause more adverse GI effects than other SSRIs currently available, which is the reason it is not recommended as a first choice. May be given as a liquid and a cap.
May give as single dose or in divided doses. Presence of food does not appreciably alter medication levels. May take up to 4-6 wk to achieve steady-state levels because of long half-life (72 h).
Long half-life is both an advantage and a drawback. If it works well, an occasional missed dose is not a problem; if problems occur, eliminating all active metabolites takes a long time. Choice depends on adverse effects and drug interactions. Adverse effects of SSRIs seem to be quite idiosyncratic; thus, if dosing is started at a conservative level and advanced as tolerated, relatively few reasons exist to recommend one over another.Adult
20 mg/d PO in morning and increase after several wk by 20 mg/d; not to exceed 80 mg/d
Note: If patient is taking 20 mg/d, may initiate once-weekly dosing with 90-mg delayed-release product 7 d after last daily dose of 20 mgPediatric
<8 years: Not established
>8 years: 10-20 mg PO qdIncreases toxicity of diazepam and trazodone by decreasing clearance; also increases toxicity of MAOIs and highly protein-bound drugs; serotonin syndrome (ie, myoclonus, rigidity, confusion, nausea, hyperthermia, autonomic instability, coma, eventual death) occurs with simultaneous use of other serotonergic agents (eg, anorectic agents, tramadol, buspirone, trazodone, clomipramine, nefazodone, tryptophan); discontinue other serotonergic agents at least 2 wk prior to SSRI initiation
Documented hypersensitivity; concurrently taking MAOIs or took them in last 2 wk; coadministration with thioridazine
Pregnancy
B – Fetal risk not confirmed in studies in humans but has been shown in some studies in animals
Precautions
Caution in hepatic impairment and history of seizures; discontinue MAOIs at least 14 d before initiating therapy
Tricyclic antidepressants
These agents are used to treat the underlying psychiatric disorder.
Doxepin (Sinequan, Zonalon)
Has antihistamine, antipruritic, and antidepressant properties. May be effective for depression (with agitation) and a primary symptom of pruritus.
Increases concentration of serotonin and norepinephrine in CNS by inhibiting their reuptake by presynaptic neuronal membrane, which is associated with a decrease in symptoms of depression.Adult
30-150 mg/d PO hs or 2-3 divided doses; gradually increase dose to 300 mg/d prn; maintain effective dose for at least 6-8 wk
Pediatric
<12 years: Not recommended
>12 years: 25-50 mg/d PO hs or bid/tid and increase gradually to 100 mg/dDecreases antihypertensive effects of clonidine but increases effects of sympathomimetics and benzodiazepines; effects of desipramine increase with phenytoin, carbamazepine, and barbiturates
Documented hypersensitivity; urinary retention; acute recovery phase following myocardial infarction; glaucoma
Pregnancy
C – Fetal risk revealed in studies in animals but not established or not studied in humans; may use if benefits outweigh risk to fetus
Precautions
Caution in cardiovascular disease, conduction disturbances, seizure disorders, urinary retention, hyperthyroidism, and thyroid replacement therapy
Amitriptyline (Elavil, Endep)
May be effective for depression with primary symptoms of pain sensations (eg, burning, chafing, stinging). Analgesia usually requires doses <50 mg qhs.
Inhibits reuptake of serotonin and/or norepinephrine at presynaptic neuronal membrane, which increases concentration in CNS. May increase or prolong neuronal activity because reuptake of these biogenic amines is important physiologically in terminating transmitting activity.Adult
25-150 mg/d mg PO hs; use minimum effective dose
Pediatric
Children: 0.1 mg/kg PO hs; increase as tolerated over 2-3 wk to 0.5-2 mg/d hs
Adolescents: 25-50 mg/d PO initially; increase gradually to 100 mg/d in divided dosesPhenobarbital may decrease effects; coadministration with CYP2D6 enzyme system inhibitors (eg, cimetidine, quinidine) may increase levels; inhibits hypotensive effects of guanethidine; may interact with thyroid medications, alcohol, CNS depressants, barbiturates, and disulfiram
Documented hypersensitivity; use of MAOIs within 14 d of initiating therapy; history of seizures, cardiac arrhythmias, glaucoma, or urinary retention
Pregnancy
C – Fetal risk revealed in studies in animals but not established or not studied in humans; may use if benefits outweigh risk to fetus
Precautions
Caution in cardiac conduction disturbances and history of hyperthyroidism, renal, impairment, or hepatic impairment; avoid use in elderly persons
Typical antipsychotics
These agents are used to treat the underlying psychiatric disorder.
Pimozide (Orap)
Previously shown to be effective for delusions of parasitosis. Centrally acting dopamine-receptor antagonist. Available in 2-mg scored tab in United States; 2-, 4-, and 10-mg tab available in Canada. Clinical response usually occurs within 10-14 d.
Adult
1-2 mg PO qd initially; increase by 2-4 mg at weekly intervals; not to exceed 10 mg/d or 200 mcg/kg/d (0.2 mg/kg/d); maintain effective therapeutic dose for at least 1 mo and then titrate down in 1-mg decrements over 1-2 wk until minimum effective dose is achieved
Pediatric
Not established
Increases toxicity of MAOIs, alfentanil, CNS depressants, and guanabenz
Documented hypersensitivity; history of cardiac arrhythmias or long QT syndrome; concurrently with macrolide antibiotics
Pregnancy
C – Fetal risk revealed in studies in animals but not established or not studied in humans; may use if benefits outweigh risk to fetus
Precautions
ECG recommended at initiation of therapy and regular intervals thereafter; careful observation for appearance of extrapyramidal symptoms (eg, akathisia, rigidity, dystonia, irreversible tardive kinesia) necessary in geriatric patients; adverse effect of restlessness (akathisia) may be treated with diphenhydramine or benztropine
Atypical antipsychotics
Lower risk of extrapyramidal adverse effects than with typical antipsychotics.
Risperidone (Risperdal)
Previously shown to be effective for delusions of parasitosis. Binds to dopamine D2 receptor with 20-times lower affinity than for 5-HT2 receptor. Improves negative symptoms of psychoses and reduces prevalence of extrapyramidal adverse effects. Indicated for treatment of psychotic disorders, including schizophrenia and bipolar disorder. Clinical response usually occurs within 10-14 d.
Adult
2-6 mg PO qhs; start at 0.5 or 1 qhs; may divide into bid dosing
Pediatric
<16 years: Not recommended
>16 years: 0.5-1 mg PO bid; not to exceed 6 mg/dCoadministration with carbamazepine may decrease effects; may inhibit effects of levodopa; clozapine may increase levels; oral solution not compatible with cola or tea
Documented hypersensitivity.
Pregnancy
C – Fetal risk revealed in studies in animals but not established or not studied in humans; may use if benefits outweigh risk to fetus
Precautions
May cause extrapyramidal reactions, hypotension, tachycardia, and arrhythmias; hyperglycemia may occur and, in some cases, be extreme, resulting in ketoacidosis, hyperosmolar coma, or death; do not split or chew oral disintegrating tab
Olanzapine (Zyprexa)
May inhibit serotonin, muscarinic, and dopamine effects. Efficacy similar to risperidone; has fewer dose-dependent adverse effects but is associated with more concern about weight gain.
Adult
5-20 mg/d PO in divided doses; lower doses recommended for dermatology-related psychoses
Pediatric
Children: Not established
Adolescents: Administer as in adultsFluvoxamine may increase effects; antihypertensives may increase risk of hypotension and orthostatic hypotension; levodopa, pergolide, bromocriptine, charcoal, carbamazepine, omeprazole, rifampin, and cigarette smoking may decrease effects
Documented hypersensitivity
Pregnancy
C – Fetal risk revealed in studies in animals but not established or not studied in humans; may use if benefits outweigh risk to fetus
Precautions
Caution in narrow-angle glaucoma, cardiovascular disease, cerebrovascular disease, prostatic hypertrophy, seizure disorders, hypovolemia, and dehydration; hyperglycemia may occur and in some cases be extreme, resulting in ketoacidosis, hyperosmolar coma, or death; administration of more than 1 IM injection is associated with substantial orthostatic hypotension (33%), thus, maintain patient in recumbent position and monitor blood pressure before repeating IM doses
Quetiapine (Seroquel)
May act by antagonizing dopamine and serotonin effects. Efficacy similar to risperidone and olanzapine. Fewer dose-dependent adverse effects and less concern for weight gain.
Adult
25 mg PO bid; titrate to 150-750 mg/d PO; not to exceed 800 mg/d; lower doses recommended for dermatology-related psychoses
Pediatric
Children: Not established
Adolescents: Administer as in adults