Cryoglobulinaemia
Cryoglobulins are single or mixed immunoglobulins that undergo reversible precipitation at low temperatures. Several types of cryoglobulins have been identified, and the potential clinical manifestations vary by cryoglobulin type.
Cryoglobulinemia is characterized by the presence of cryoglobulins in the serum. This may result in a clinical syndrome of systemic inflammation (most commonly affecting the kidneys and skin) caused by cryoglobulin-containing immune complexes.
Cryoglobulinemia may be classified based on cryoglobulin composition with the Brouet classification, which is as follows:
- Type I cryoglobulinemia, or simple cryoglobulinemia, is the result of a monoclonal immunoglobulin, usually immunoglobulin M (IgM) or, less frequently, immunoglobulin G (IgG), immunoglobulin A (IgA), or light chains.
- Types II and III cryoglobulinemia (mixed cryoglobulinemia) contain rheumatoid factors (RFs), which are usually IgM and, rarely, IgG or IgA. These RFs form complexes with the fragment, crystallizable (Fc) portion of polyclonal IgG. The actual RF may be monoclonal (in type II cryoglobulinemia) or polyclonal (in type III cryoglobulinemia) immunoglobulin. Types II and III cryoglobulinemia represent 80% of all cryoglobulins.
Cryoglobulinemia may also be classified based on the association of the syndrome with an underlying disease. Cryoglobulinemia without an associated disease has been known as essential, or idiopathic, cryoglobulinemia. However, the discovery of a close association between hepatitis C virus (HCV) and mixed cryoglobulinemia has cast doubt on the existence of essential, or idiopathic, cryoglobulinemia.1 Cryoglobulinemia associated with a particular disease (lymphoproliferative disorder, autoimmune disease, infectious disease) is known as secondary cryoglobulinemia.
Pathophysiology
The mechanisms of cryoprecipitation are poorly understood, but several factors have been investigated. The solubility of cryoglobulins has been found to be partially related to the structure of component immunoglobulin heavy and light chains.2,3,4 Alteration in protein conformation with temperature changes also leads to decreased solubility and subsequent vasculitic damage.5,6 The ratio of antibody to antigen in circulating cryoglobulin aggregates or immune complexes affects the rate of clearance from the circulation and the resultant rate and location of tissue deposition.7
Some of the sequelae of cryoglobulinemia are thought to be related to immune-complex disease (eg, glomerulonephritis, chronic vasculitis), but not all persons with cryoglobulinemia present with these manifestations. Individuals with cryoglobulinemia may have intravascular cryoglobulin deposits, a reduced level of complement, and complement fragments (C3a, C5a) that act as chemotactic mediators of inflammation; however, the pathophysiologic process of this disease has not been fully explained. Other sequelae are directly related to cryoprecipitation in vivo, including plugging and thrombosis of small arteries and capillaries in the extremities (gangrene) and glomeruli (acute renal failure). Circulating large–molecular-weight cryoprotein complexes, even when unprecipitated in vivo, can lead to clinical hyperviscosity syndrome.
Type I cryoglobulins are usually monoclonal IgM and, less frequently, IgG, IgA, or light chains. Type I cryoglobulins rarely have RF activity and do not activate complement in vitro. This disorder is typically related to an underlying lymphoproliferative disease and, as such, may be clinically indistinguishable from Waldenström macroglobulinemia, multiple myeloma, or chronic lymphocytic leukemia. Type I cryoglobulinemia may result in hyperviscosity due to high levels of circulating monoclonal cryoglobulin, leading to physical obstruction of vessels. Concentrations may reach up to 8 g/L. In addition, nonobstructive damage may be mediated by immune complex deposition and subsequent inflammatory vasculitis.
Types II and III, also known as the mixed cryoglobulinemias, are associated with chronic inflammatory states such as systemic lupus erythematosus (SLE), Sjögren syndrome, and viral infections (particularly HCV). In these disorders, the IgG fraction is always polyclonal with either monoclonal (type II) or polyclonal (type III) IgM (rarely IgA or IgG) with RF activity (ability to bind IgG). B-cell clonal expansion, particularly RF-secreting cells, is a distinctive feature in many of these disease states.1,8,9,10
The resultant aggregates and immune complexes are thought to outstrip reticuloendothelial-clearing activity. Tissue damage results from immune complex deposition and complement activation. Of note, in HCV-related disease, HCV-related proteins are thought to play a direct role in pathogenesis and are present in damaged skin, blood vessels, and kidneys
- Specific clinical manifestations associated with type I cryoglobulinemia are related to hyperviscosity and thrombosis, as would be expected given their usual high concentrations of immunoglobulins and limited interference with complement function. These manifestations include acrocyanosis, retinal hemorrhage, severe Raynaud phenomenon with digital ulceration, livedo reticularis, purpura, and arterial thrombosis.
- Specific clinical manifestations associated with types II and III cryoglobulinemia include joint involvement (usually, arthralgias in the proximal interphalangeal [PIP] joints, metacarpophalangeal [MCP] joints, knees, and ankles), fatigue, myalgias, renal immune-complex disease, cutaneous vasculitis, and peripheral neuropathy.
- Typical presentations and reported frequencies include the following:
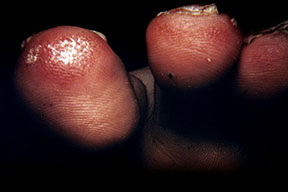
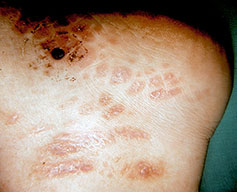
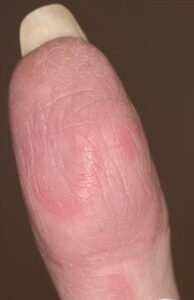
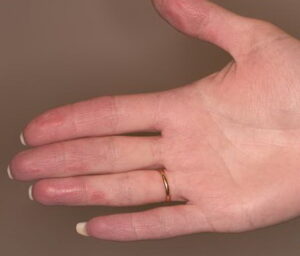
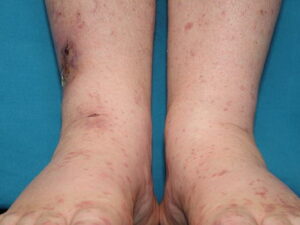
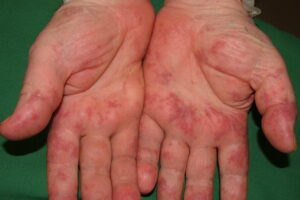
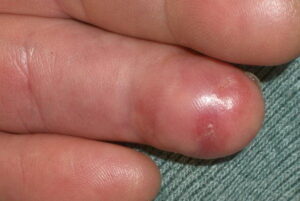
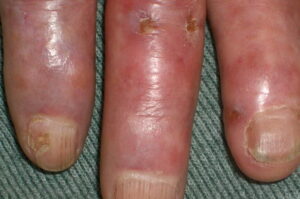
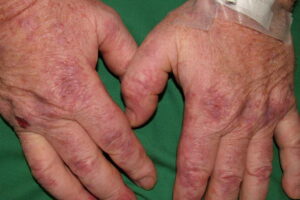
- Cutaneous: These manifestations are nearly always present in cryoglobulinemia. Observed lesions have a predilection for dependent areas (particularly the lower extremities) and include erythematous macules and purpuric papules (90-95%), as well as ulcerations (10-25%).15,21,14,22 Lesions in nondependent areas are more common in type I cryoglobulinemia (head and mucosa), as are livedo reticularis, Raynaud phenomenon, and ulcerations. Nailfold capillary abnormalities are common and include dilatation, altered orientation, capillary shortening, and neoangiogenesis.
- Musculoskeletal: Symptoms such as arthralgias and myalgias are rare in type I cryoglobulinemia and are common in types II and III disease. Frank arthritis and myositis are rare. Arthralgias commonly affect the proximal interphalangeal and metacarpophalangeal joints of the hands, knees, and ankles. Musculoskeletal symptoms are described in more than 70% of persons with cryoglobulinemia.24,25,21
- Renal: Renal disease may occur secondary to thrombosis (type I cryoglobulinemia) or immune complex deposition (types II and III). The incidence of renal disease varies from 5-60%. Histologically, membranoproliferative glomerulonephritis is almost always the lesion in mixed cryoglobulinemia. Clinically, isolated proteinuria and hematuria are more common than nephrotic syndrome, nephritic syndrome, or acute renal failure. Renal involvement is one of the most serious complications of cryoglobulinemia and typically manifests early in the course of the disease (within 3-5 y of diagnosis). Failure to treat may result in renal failure.15,26,27
- Pulmonary: A reduction in forced expiratory flow rates and the presence of interstitial infiltrates revealed by chest radiographs are common in mixed cryoglobulinemia. Approximately 40-50% of patients are symptomatic with dyspnea, cough, or pleuritic pain. Severe pulmonary disease is rare.28,29,30,31
- Neuropathy: Neuropathy is common in types II and III disease (as determined with electromyographic and nerve conduction studies), affecting 70-80% of patients. Symptomatic disease was once reported as less common (5-40%); however, more recently, subjective symptoms have been reported up to 91% of patients. Sensory fibers are more commonly affected than motor fibers, with pure motor neuropathy in approximately 5% of patients.32,21,33,34
- Abdominal pain: Abdominal pain has been reported in 2-22% of patients. Vasculitis of the small mesenteric vessels that leads to acute abdomen has been reported.
- Sicca symptoms have been reported in 4-20% of patients.21,28
- Acrocyanosis has been reported in up to 9% of patients.
- Arterial thrombosis has been reported in 1% of patients.
- Meltzer triad, ie, purpura, arthralgia, and weakness, was first described in 1966 by Meltzer and Franklin in cases of essential mixed cryoglobulinemia. This triad is generally seen with types II and III cryoglobulinemia and is seen in up to 25-30% of patients.35,21
Physical
- Skin manifestations
- Ischemic necrosis (40% in type I, 0-20% in mixed types)
- Palpable purpura (15% in type I, 80% in mixed types)
- Livedoid vasculitis (1% in type I, 14% in type III)
- Cold-induced urticaria (15% in type I, 10% in type III)
- Hyperkeratotic spicules in areas exposed to cold
- Scarring of tip of nose, pinnae, fingertips, and toes
- Acrocyanosis
- Nailfold capillary abnormalities
- Pulmonary manifestations
- Dyspnea
- Cough
- Pleurisy
- Pleural effusions
- Bronchiectasis
- Gastrointestinal manifestations
- Abdominal pain (2-22%)
- Hemorrhage
- Hepatomegaly or signs of cirrhosis (ie, palmar erythema, abdominal wall collateral vessels, spider angiomata)
- Splenomegaly
- Renal manifestations
- Membranoproliferative glomerulonephritis described in all types (more common in type II)
- Intraluminal cryoglobulin deposition
- Hypertension
- Nephrotic-range proteinuria with resultant edema
- Joint manifestations
- Arthralgias (5% of type I, 20-58% of mixed)
- Frank arthritis and progressive joint deformity (distinctly rare)
- Nervous system manifestations
- Sensorimotor neuropathy
- Visual disturbances
- CNS involvement (rare, although pseudotumor cerebri and cerebral vascular events have been described)
- Other manifestations – Fever
Causes
- Disease associations variable based on type of cryoglobulinemia
- Type I is observed in lymphoproliferative disorders (eg, multiple myeloma, Waldenström macroglobulinemia).
- Types II and III are observed in chronic inflammatory diseases such as chronic liver disease, infections (chronic HCV infection), and coexistent connective-tissue diseases (SLE, Sjögren syndrome). Mixed cryoglobulinemia is rarely associated with lymphoproliferative disorders.
- Infection
- Viral – Hepatitis A, B, and C (see Differentials); HIV; Epstein-Barr virus (EBV); cytomegalovirus (CMV); adenovirus; chikungunya
- Bacterial – Endocarditis, streptococcal infections, syphilis, Lyme disease, leprosy, Q fever, brucellosis
- Fungal -Coccidioidomycosis
- Parasitic -Malaria, toxoplasmosis, others
- Autoimmune diseases
- SLE, rheumatoid arthritis, Sjögren syndrome
- Vasculitis -Polyarteritis nodosa (especially hepatitis B–associated), Henoch-Schönlein purpura
- Lymphoproliferative disorders – Waldenström macroglobulinemia, multiple myeloma, lymphoma, leukemia (eg, chronic lymphocytic leukemia, hairy cell leukemia)
- Renal diseases -Proliferative glomerulonephritis
- Liver diseases – Hepatitis A, B, and C (30-98% of patients with HCV infection have cryoglobulins, especially type II); cirrhosis
- Familial
- Essential
- Experimental – Postvaccination (eg, pneumococcal vaccine)
Laboratory Studies
- Evaluation for serum cryoglobulins
- The blood specimen must be obtained in warm tubes (37°C) in the absence of anticoagulants.
- Allow the blood sample to clot before removal of serum with centrifugation (at 37°C).
- The period required for the serum sample to incubate (at 4°C) depends on the type of cryoglobulin present, as follows:
- Type I tends to precipitate within the first 24 hours (at concentrations >5 mg/mL).
- Type III cryoglobulins may require 7 days to precipitate a small sample (<1 mg/mL).
- Repeat centrifugation to determine cryocrit (volume of precipitate as a percentage of original serum volume).
- Cryoglobulin concentration may be determined via spectrophotometric analysis. Specific immunologic assays may be used to identify cryoglobulin components (immunoglobulins, light chains, clonality).
- Urinalysis: Abnormalities may represent evidence of renal disease.
- Complete blood cell count: Leukocytosis may be a manifestation of concomitant infection or leukemia. Anemia may be present.
- Serum chemistry: Patients with renal insufficiency may present with elevated serum creatinine levels and electrolyte abnormalities.
- Liver function studies: Liver function studies may reveal evidence of underlying hepatitis; obtain hepatitis serology.
- RF: RF is positive in types II and III.
- Antinuclear antibody (ANA): ANA is indicated upon clinical suspicion of underlying connective-tissue disease (SLE, Sjögren syndrome).
- Erythrocyte sedimentation rate (ESR): Elevations may be secondary to rouleaux formation.
- Complement evaluation (CH50, C3, C4): Patients may display hypocomplementemia (especially low C4 levels).
- Other studies: Consider serum protein electrophoresis (SPEP), urine protein electrophoresis (UPEP), and quantitative immunoglobulin upon suspicion for underlying gammopathy.
- Serum viscosity: Measure serum viscosity if symptoms warrant.
- Further diagnostic laboratory tests: Consider further testing based on the level of suspicion for other associated disease. For example, a recent study demonstrated that patients with mixed cryoglobulinemia associated with HCV infection have elevated levels of interferon-inducible protein 10 and that these levels correlate with disease activity.36
Imaging Studies
- A chest radiograph may reveal interstitial involvement or pleural effusions.
- CT imaging may be considered upon high suspicion of underlying malignancy.
- Transesophageal echocardiography should be obtained if bacterial endocarditis is suspected.
- Angiography may be considered to evaluate for evidence of vasculitis.
Procedures
- Tissue biopsy may be required for diagnosis when patients with vasculitis, renal disease, or both are evaluated.
- Electromyography and nerve conduction studies may be used to confirm neuropathy when history or physical examination findings are suggestive.
- Further diagnostic procedures (eg, bone marrow biopsy, liver or kidney biopsy) usually depend on coexistent disease, especially HCV infection.
Histologic Findings
Skin: Purpura are histologically characterized by dermal vasculitis that extends variably to the subcutaneous interstitial space. HCV-associated proteins have been found in vasculitic skin biopsy samples, suggesting a role for these antigens in pathogenesis of the lesions.
Other organs: Autopsy studies have revealed unsuspected vasculitis of multiple organs (heart, lung, gastrointestinal tract, central nervous system, liver, muscle, adrenals).14 Histologic evaluation of affected lung, kidney, and muscle reveals eosinophilic material in the lumen of small vessels with frequent extension into the vessel intima and inflammation of the vessel wall.37
Although biopsy samples generally exhibit inflammatory vascular changes (eg, leukocytoclastic vasculitis in patients with vasculitic purpura), intraluminal cryoglobulin deposits may be observed, especially in renal glomeruli
Medical Care
The goal of therapy is to treat underlying conditions, as well as to limit the precipitant cryoglobulin and the resultant inflammatory effects. Thus, HCV testing is required. HCV-antibody or HCV-RNA testing may be diagnostic. If HCV test results are negative and clinical suspicion remains high, these tests may be performed on the cryoprecipitate. Asymptomatic cryoglobulinemia does not require treatment. Some authors recommend intervening as little as possible except when faced with severe deterioration of renal or neurologic function. Secondary cryoglobulinemia is best managed with treatment of the underlying malignancy or associated disease. Otherwise, cryoglobulinemia is treated simply with suppression of the immune response. A paucity of controlled studies evaluating the relative efficacy of various therapies limits the use of existing data.
- Nonsteroidal anti-inflammatory drugs (NSAIDs) may be used in patients with arthralgia and fatigue.
- Immunosuppressive medications (eg, corticosteroid therapy and/or cyclophosphamide or azathioprine) are indicated upon evidence of organ involvement such as vasculitis, renal disease, progressive neurologic findings, or disabling skin manifestations.
- Plasmapheresis is indicated for severe or life-threatening complications related to in vivo cryoprecipitation or serum hyperviscosity. Concomitant use of high-dose corticosteroids and cytotoxic agents is recommended for reduction of immunoglobulin production. Some authors recommend using concomitant cytotoxic medications or corticosteroids to reduce a rebound phenomenon that may develop after plasmapheresis.
- Pegylated interferon alfa (IFN-alfa) combined with ribavirin has demonstrated efficacy in patients with cryoglobulinemia associated with hepatitis C, and efficacy in patients with chronic myelogenous leukemias and low-grade lymphomas has been reported. The details of therapy and the recommended approach vary based on the clinical setting, and expert opinion should be sought.
- Case reports have detailed the remission of hepatitis B–related cryoglobulinemic vasculitis with entecavir therapy.38
- Small and uncontrolled studies suggest the anti-CD20 chimeric monoclonal antibody rituximab is effective in controlling disease manifestations such as vasculitis, peripheral neuropathy, arthralgias, low-grade B-cell lymphomas, renal disease, and fever.39,40 Rituximab therapy has been used predominately in HCV-related mixed cryoglobulinemia refractory to or unsuitable for corticosteroids and antiviral (IFN-alfa) therapy. Rituximab therapy is reportedly well tolerated in this patient population; however, treatment results in increased titers of HCV-RNA of undetermined significance. The National Institutes of Health has launched a large trial of rituximab in the treatment of mixed cryoglobulinemia.
Consultations
- Rheumatologist or clinical immunologist
- Nephrologist upon evidence of renal disease (ie, hypertension, abnormal findings on urinalysis)
- Hematologist upon evidence of underlying hematological disease or for plasmapheresis
- Gastroenterologist or hepatologist for patients with underlying hepatitis
Medication
The overall aim of therapy is treatment of any underlying condition and general suppression of the immune response. Mild anti-inflammatory medications (eg, NSAIDs) are effective in mild cases, and corticosteroid therapy is reserved for the more severe or refractory cases. Patients who require potent immunosuppression or other more aggressive therapies for severe disease should be treated by a specialist. Cyclophosphamide may be used as a steroid-sparing agent or administered concomitantly in severe cases of vasculitis, particularly in patients with renal disease. Azathioprine is commonly used as a steroid-sparing agent, and chlorambucil has also been used for severe vasculitis.
Nonsteroidal anti-inflammatory drugs
NSAIDs such as ibuprofen, naproxen, and indomethacin have analgesic, anti-inflammatory, and antipyretic activities. Their mechanism of action is not known, but they may inhibit cyclooxygenase activity and prostaglandin synthesis. NSAIDs may have additional mechanisms, such as inhibition of leukotriene synthesis, lysosomal enzyme release, lipoxygenase activity, neutrophil aggregation, and various cell membrane functions. NSAIDs are used to reduce the resultant inflammatory response of cryoglobulin precipitation.
Ibuprofen (Advil, Motrin, Excedrin IB, Ibuprin)
NSAIDs are the DOC in patients with mild symptoms of arthralgia or fatigue. Inhibits inflammatory reactions and pain by decreasing prostaglandin synthesis.
- Dosing
- Interactions
- Contraindications
- Precautions
Adult
600-800 mg PO tid/qid
Pediatric
30-40 mg/kg/d PO divided tid/qid
- Dosing
- Interactions
- Contraindications
- Precautions
Coadministration with aspirin increases the risk of inducing serious NSAID-related adverse effects; probenecid may increase concentrations and, possibly, toxicity of NSAIDs; may decrease effect of hydralazine, ACE inhibitors, angiotensin II receptor blockers, and beta-blockers; may decrease diuretic effects of furosemide and thiazides; coadministration with ACE inhibitors, angiotensin II receptor blockers, and potassium-sparing diuretics may result in hyperkalemia; may increase PT when combined with anticoagulants or aggravate bleeding tendency because of the antiplatelet effect of NSAIDs; phenytoin levels may be increased when administered concurrently
- Dosing
- Interactions
- Contraindications
- Precautions
Documented hypersensitivity; peptic ulcer disease; history of GI bleeding or perforation; renal insufficiency; anticoagulation; coagulopathy
- Dosing
- Interactions
- Contraindications
- Precautions
Pregnancy
B – Fetal risk not confirmed in studies in humans but has been shown in some studies in animals
D – Fetal risk shown in humans; use only if benefits outweigh risk to fetus
Precautions
Most common toxicities include gastrointestinal manifestations such as nausea, abdominal pain, peptic ulcer disease, and renal insufficiency; may cause increased blood pressure in patients with hypertension due to blunting of effects of antihypertensive medications; patients with congestive heart failure may have exacerbations due to fluid and sodium retention; caution in coagulation abnormalities or during anticoagulant therapy
Corticosteroids
These medications are used to reduce the resultant immune response from cryoglobulin precipitation, particularly in patients with more severe symptoms or some evidence of organ damage
Prednisone (Sterapred)
DOC in patients with evidence of acute vasculitis.
- Dosing
- Interactions
- Contraindications
- Precautions
Adult
1 mg/kg/d PO in divided doses; up to 120 mg/d has been reported
Pediatric
2-3 mg/kg/d PO
- Dosing
- Interactions
- Contraindications
- Precautions
Coadministration with estrogens may decrease prednisone clearance; concurrent use with digoxin may cause digitalis toxicity secondary to hypokalemia; phenobarbital, phenytoin, and rifampin may increase metabolism of glucocorticoids (consider increasing maintenance dose); monitor for hypokalemia with coadministration of diuretics
- Dosing
- Interactions
- Contraindications
- Precautions
Documented hypersensitivity
- Dosing
- Interactions
- Contraindications
- Precautions
Pregnancy
B – Fetal risk not confirmed in studies in humans but has been shown in some studies in animals
Precautions
Caution in severe bacterial, viral, or fungal infection; active peptic ulcer disease; diabetes mellitus; toxicities include weight gain, dyspepsia, mood changes, infection, peptic ulcer disease, hypertension, diabetes mellitus, osteoporosis, avascular necrosis, cataracts, glaucoma, myopathy, and skin changes; growth retardation in children; abrupt discontinuation may result in adrenal crisis
Immunosuppressive agents
These are commonly used as steroid-sparing agents.
Cyclophosphamide (Cytoxan, Neosar)
Chemically related to nitrogen mustards. As an alkylating agent, the mechanism of action of the active metabolites may involve cross-linking of DNA, interfering with growth of normal and neoplastic cells.
- Dosing
- Interactions
- Contraindications
- Precautions
Adult
1-5 mg/kg/d PO
Pediatric
Administer as in adults
- Dosing
- Interactions
- Contraindications
- Precautions
Allopurinol may increase risk of bleeding or infection and enhance myelosuppressive effects; may potentiate doxorubicin-induced cardiotoxicity; may reduce digoxin serum levels and antimicrobial effects of quinolones; chloramphenicol may increase half-life while decreasing metabolite concentrations; may increase effect of anticoagulants; coadministration with high doses of phenobarbital may increase rate of metabolism and leukopenic activity; thiazide diuretics may prolong cyclophosphamide-induced leukopenia and neuromuscular blockade by inhibiting cholinesterase activity
- Dosing
- Interactions
- Contraindications
- Precautions
Documented hypersensitivity; infection; severely depressed bone marrow function; severe cytopenias
- Dosing
- Interactions
- Contraindications
- Precautions
Pregnancy
D – Fetal risk shown in humans; use only if benefits outweigh risk to fetus
Precautions
Toxicities include nausea and vomiting, leukopenia, thrombocytopenia, anemia, infection, alopecia, hemorrhagic cystitis, infertility, teratogenicity, and increased risk of infection; monitor CBC count and UA at regular intervals
Azathioprine (Imuran)
Antagonizes purine metabolism and inhibits synthesis of DNA, RNA, and proteins. May decrease proliferation of immune cells, which results in lower autoimmune activity.
- Dosing
- Interactions
- Contraindications
- Precautions
Adult
2-3 mg/kg/d PO single or divided dose
1 mg/kg/d initial; increase depending on clinical and hematologic response and toxicity
Pediatric
Administer as in adults
- Dosing
- Interactions
- Contraindications
- Precautions
Allopurinol may increase risk of bleeding or infection and enhance myelosuppressive effects; toxicity increases with allopurinol; concurrent use with ACE inhibitors may induce severe leukopenia; may increase levels of methotrexate metabolites and decrease effects of anticoagulants, neuromuscular blockers, and cyclosporine
- Dosing
- Interactions
- Contraindications
- Precautions
Documented hypersensitivity; low levels of serum thiopurine methyltransferase (TPMT); active infection (relative); severe cytopenias (relative)
- Dosing
- Interactions
- Contraindications
- Precautions
Pregnancy
B – Fetal risk not confirmed in studies in humans but has been shown in some studies in animals
Precautions
Nausea and vomiting; hematologic toxicities may occur; check TPMT level prior to initiation of therapy; pancreatitis rarely occurs; monitor CBC count and LFTs at regular intervals; may also monitor 6-thioguanine (6-TG) and 6-methyl mercaptopurine (6-MMP) levels
Chlorambucil (Leukeran)
Alkylates and cross-links strands of DNA, inhibiting DNA replication and RNA transcription.
- Dosing
- Interactions
- Contraindications
- Precautions
Adult
0.1-0.2 mg/kg/d PO
Pediatric
Administer as in adults
- Dosing
- Interactions
- Contraindications
- Precautions
None reported
- Dosing
- Interactions
- Contraindications
- Precautions
Documented hypersensitivity; previous resistance to medication; active infection (relative)
- Dosing
- Interactions
- Contraindications
- Precautions
Pregnancy
D – Fetal risk shown in humans; use only if benefits outweigh risk to fetus
Precautions
Caution in history of seizure disorders or in patients diagnosed with bone marrow suppression; gastrointestinal effects, dermatitis or erythema multiforme, cystitis, pulmonary fibrosis, hepatotoxicity, infertility, teratogenic effects, peripheral neuropathy, and secondary malignancy may occur; monitor CBC count and LFTs at regular intervals
Interferons
These agents are naturally produced proteins with antiviral, antitumor, and immunomodulatory actions. IFN-alfa is generally administered subcutaneously.
Interferon alfa-2b (Intron A)
Protein product manufactured by recombinant DNA technology. Mechanism of antitumor activity is not clearly understood; however, direct antiproliferative effects against malignant cells and modulation of host immune response may play important roles. Has antiviral activity in HCV infection.
- Dosing
- Interactions
- Contraindications
- Precautions
Adult
Not recommended without consultation
Pediatric
Not established
- Dosing
- Interactions
- Contraindications
- Precautions
Potential risk of renal failure when administered concurrently with interleukin-2; theophylline may increase IFN-alfa toxicity by reducing clearance; cimetidine may increase antitumor effects of IFN-alfa; zidovudine and vinblastine may increase toxicity of IFN-alfa
- Dosing
- Interactions
- Contraindications
- Precautions
Documented hypersensitivity; patients who have anaphylactic sensitivity to mouse IgG, egg protein, or neomycin; autoimmune disease (relative)
- Dosing
- Interactions
- Contraindications
- Precautions
Pregnancy
C – Fetal risk revealed in studies in animals but not established or not studied in humans; may use if benefits outweigh risk to fetus
Precautions
Fatigue; headache; arthralgias; myalgias; fever; nausea; autoimmunity; depression and suicidal ideation may be adverse effects of treatment; infrequently, severe or fatal GI hemorrhage is reported in association with IFN-a therapy; prior to initiation of therapy, perform tests to quantitate peripheral blood hemoglobin, platelets, granulocytes, hairy cell, and bone marrow hairy cells; monitor periodically (eg, monthly) during treatment to determine response to treatment; if patient does not respond within 6 mo, discontinue treatment; if a response occurs, continue treatment until no further improvement is observed; whether continued treatment after that time is beneficial is not known
Peginterferon alfa-2a (Pegasys)
Used in combination with ribavirin to treat patient with chronic HCV infection who have compensated liver disease and have not received IFN-alfa previously. Consists of interferon alfa-2a attached to a 40-kD branched PEG molecule. Predominantly metabolized by the liver.
- Dosing
- Interactions
- Contraindications
- Precautions
Adult
180 mcg SC qwk
Pediatric
Not established
- Dosing
- Interactions
- Contraindications
- Precautions
Theophylline may increase toxicity by reducing clearance; cimetidine may increase the antitumor effects; zidovudine and vinblastine may increase toxicity
- Dosing
- Interactions
- Contraindications
- Precautions
Documented hypersensitivity; decompensated liver disease; significant preexisting psychiatric disease; ongoing or recent alcohol use; platelet count <70,000/µL
- Dosing
- Interactions
- Contraindications
- Precautions
Pregnancy
C – Fetal risk revealed in studies in animals but not established or not studied in humans; may use if benefits outweigh risk to fetus
Precautions
Insomnia; mental dysfunction (eg, mood dysfunction, depression, psychosis, aggressive behavior, hallucinations, violent behavior, suicidal ideation, suicide attempt, suicide, homicidal ideation [rare]), even without previous history of psychiatric illness; flulike symptoms; rash and pruritus; anorexia; neutropenia; thrombocytopenia; thyroid dysfunction; retinal abnormalities
Peginterferon alfa 2b (PEG-Intron)
Escherichia coli recombinant product. Used to treat chronic HCV infection in patients not previously treated with INF-alfa who have compensated liver disease. Exerts cellular activities by binding to specific membrane receptors on cell surface, which, in turn, may suppress cell proliferation and may enhance phagocytic activity of macrophages. May also increase cytotoxicity of lymphocytes for target cells and inhibit virus replication in virus-infected cells.
- Dosing
- Interactions
- Contraindications
- Precautions
Adult
Inject SC qwk for 1 y using weight-based dosing as follows:
37-45 kg: 40 mcg (0.4 mL of 100 mcg/mL)
46-56 kg: 50 mcg (0.5 mL of 100 mcg/mL)
57-72 kg: 64 mcg (0.4 mL of 160 mcg/mL)
73-88 kg: 80 mcg (0.5 mL of 160 mcg/mL)
89-106 kg: 96 mcg (0.4 mL of 240 mcg/mL)
107-136 kg: 120 mcg (0.5 mL of 240 mcg/mL)
137-160 kg: 150 mcg (0.5 mL of 300 mcg/mL)
Pediatric
Not established
- Dosing
- Interactions
- Contraindications
- Precautions
Concurrent administration with interleukin 2 may increase nephrotoxicity; theophylline, zidovudine, and vinblastine may increase toxicity; cimetidine may increase antitumor effects
- Dosing
- Interactions
- Contraindications
- Precautions
Documented hypersensitivity; autoimmune hepatitis; pancreatitis; colitis
- Dosing
- Interactions
- Contraindications
- Precautions
Pregnancy
C – Fetal risk revealed in studies in animals but not established or not studied in humans; may use if benefits outweigh risk to fetus
Precautions
Insomnia; mental dysfunction (eg, mood dysfunction, depression, psychosis, aggressive behavior, hallucinations, violent behavior, suicidal ideation, suicide attempt, suicide, homicidal ideation [rare]), even without previous history of psychiatric illness; flulike symptoms; rash and pruritus; anorexia; neutropenia; thrombocytopenia; thyroid dysfunction; retinal abnormalities
Antiviral agents
Nucleoside analogs are initially phosphorylated by viral thymidine kinase to eventually form a nucleoside triphosphate. These molecules inhibit herpes simplex virus (HSV) polymerase with 30-50 times the potency of human alpha-DNA polymerase.
Ribavirin (Virazole)
Antiviral nucleoside analogs. Chemical name is 1-beta-D-ribofuranosyl-1H-1,2,4-triazole-3-carboxamide. Given alone, has little effect on the course of HCV infection. When used with IFN, significantly augments rate of sustained virologic response.
- Dosing
- Interactions
- Contraindications
- Precautions
Adult
10.6 mg/kg/d PO or divided bid
Pediatric
Not established
- Dosing
- Interactions
- Contraindications
- Precautions
Decreases zidovudine effects
- Dosing
- Interactions
- Contraindications
- Precautions
Documented hypersensitivity
- Dosing
- Interactions
- Contraindications
- Precautions
Pregnancy
X – Contraindicated; benefit does not outweigh risk
Precautions
Closely monitor patients with COPD and asthma for deterioration of respiratory function
Antiviral Agent, Oral
This agent inhibits the viral reverse transcriptase enzyme, which limits viral replication.
Entecavir (Baraclude)
Guanosine nucleoside analogue with activity against HBV polymerase. Competes with natural substrate deoxyguanosine triphosphate to inhibit HBV polymerase activity (ie, reverse transcriptase). Less effective for lamivudine-refractory HBV infection. Indicated for treatment of chronic hepatitis B infection. Available as tab and oral solution (0.05 mg/mL; 0.5 mg = 10 mL).
- Dosing
- Interactions
- Contraindications
- Precautions
Adult
Treatment for nucleoside naive: 0.5 mg PO qd 2 h ac or 2 h pc
CrCl 30-49 mL/min: 0.25 mg PO qd or 0.5 mg q48h
CrCl 10-29 mL/min: 0.15 mg PO qd or 0.5 mg q72h
CrCl <10 mL/min: 0.05 mg PO qd or 0.5 mg qwk
Receiving lamivudine or lamivudine resistance: 1 mg PO qd 2 h ac or 2 h pc
CrCl 30-49 mL/min: 0.5 mg PO qd or 1 mg q48h
CrCl 10-29 mL/min: 0.3 mg PO qd or 1 mg q72h
CrCl <10 mL/min: 0.1 mg PO qd or 1 mg qwk
Pediatric
<16 years: Not established
>16 years: Administer as in adults
- Dosing
- Interactions
- Contraindications
- Precautions
Not a substrate, inhibitor, or inducer of cytochrome P450; coadministration with drugs that reduce renal function (eg, aminoglycosides, cidofovir, cyclosporine) or that compete for active tubular secretion (eg, probenecid, salicylates) may increase serum concentration of either entecavir or coadministered drug
- Dosing
- Interactions
- Contraindications
- Precautions
Documented hypersensitivity
- Dosing
- Interactions
- Contraindications
- Precautions
Pregnancy
C – Fetal risk revealed in studies in animals but not established or not studied in humans; may use if benefits outweigh risk to fetus
Precautions
Reduce dose with renal impairment; if on hemodialysis, administer afterwards; common adverse effects include headache, tiredness, dizziness, and nausea; may elevate liver enzyme levels; may cause lactic acidosis; severe acute exacerbations of hepatitis B may occur in patients who discontinue antihepatitis B therapy
Antineoplastic agents
These agents inhibit cell growth and proliferation.
Rituximab (Rituxan)
Genetically engineered human monoclonal antibody directed against the CD20 antigen found on the surface of normal and malignant B lymphocytes.
Immunomodulates response against malignant cells.
- Dosing
- Interactions
- Contraindications
- Precautions
Adult
60-75 mg/m2 IV as a single dose; repeat q21d
Alternatively, 20-30 mg/m2/d for 2-3 d; repeat in 4 wk
Pediatric
Not established