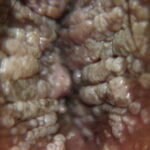
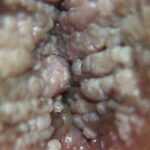
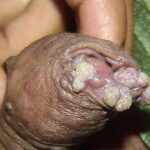
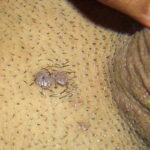
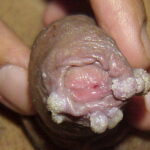
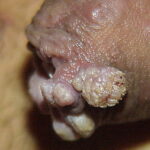
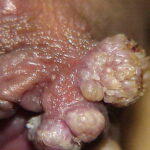
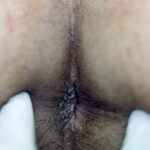
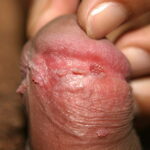
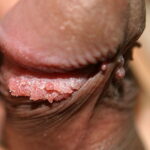
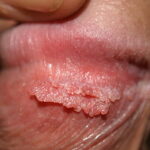
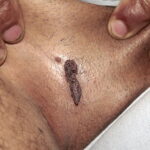
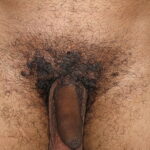
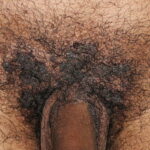
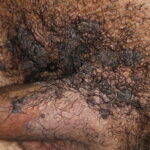
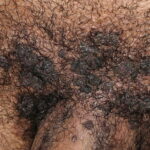
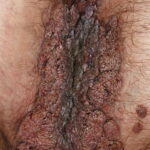
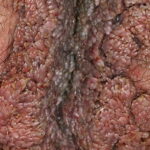
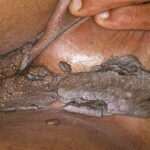
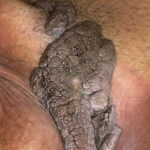
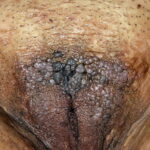
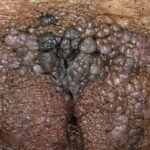
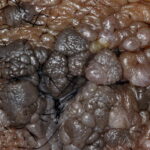
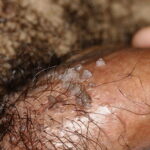
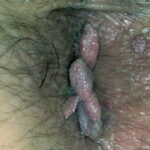
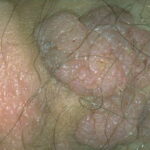
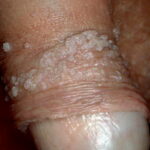
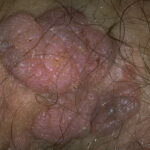
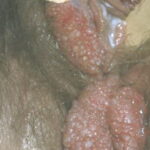
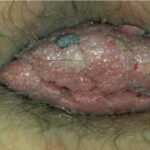
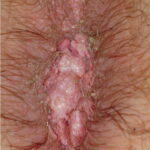
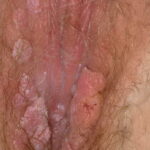
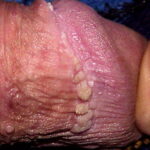
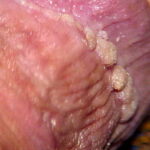
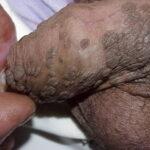
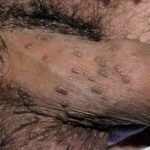
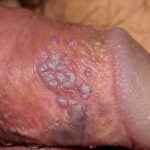
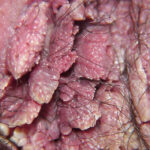
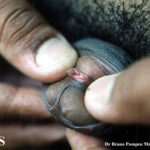
Condyloma acuminatum
The viral nature of genital warts was first recognized in 1907 when Ciuffo induced warts after autoinoculation of cell-free wart extracts.1 The human papillomavirus (HPV) was identified with the development of molecular biology techniques as the virus responsible for condyloma acuminatum. Zur Hansen proposed that HPV was likely important in the etiology of genital tract neoplasias in the mid 1970s.2 The DNA of the first genital wart was characterized in 1980. Today, more than 120 distinct HPV subtypes have been identified. This group of viruses is strongly linked to the development of cervical cancer. HPV contributes to 90% of anal cancers and 40% of vulva, vaginal, and penile cancers. Squamous cell carcinoma of the oropharynx is associated with HPV in 50% of cases.3,4
Complete understanding of the natural history of HPV disease has significantly improved over the last 20 years, but key issues remain unanswered. Topics requiring further research include HPV age-specific outcomes, risk of progression and regression of disease, and factors important in the acquisition of immunity following infection.5
Genital warts are transmitted by sexual contact. Approximately two thirds of individuals who have sexual contact with an infected partner develop genital warts. The exact incubation time is unknown, but most investigators believe the incubation period is 3 weeks to 8 months.6,7
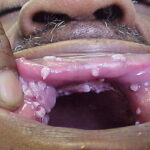
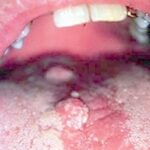
HPV is a group of double-stranded DNA viruses. The genome encodes 6 early open reading frames (E1, E2, E4, E5, E6, E7) and 2 late open reading frames (L1, L2). The E genes encode proteins important in regulatory function, and the L genes encode for viral capsid proteins. This group of viruses can infect many different sites, including the larynx, skin, mouth, esophagus, and the anogenital tract.
Approximately 40 different types of HPV can infect the anogenital tract. Infection caused by the HPV virus results in local infections and appears as warty papillary condylomatous lesions. HPV infections in the genital area are sexually transmitted.
History
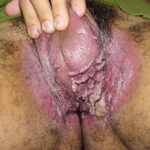
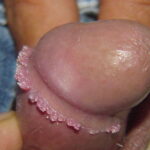
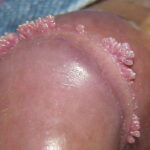
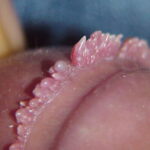
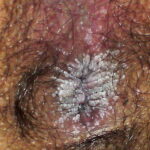
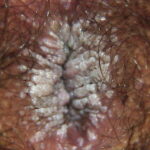
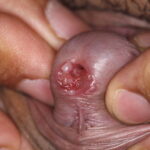
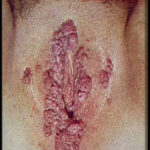
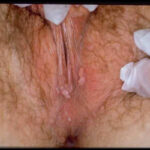
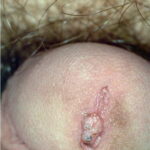
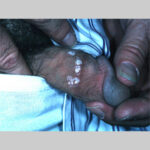
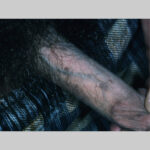
Most patients seek medical care when they notice lumps on the vulva, perianal area, or periclitoral area.
- These lesions are generally not painful, but they can be associated with pruritus.
- Bleeding can be observed if the lesions become confluent and are irritated by clothing.
Physical
- Inspection of the female genital area requires good lighting.
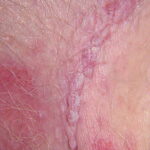
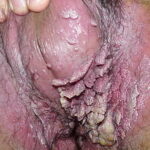
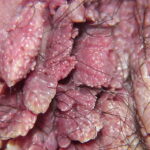
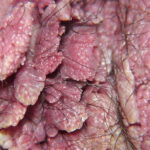
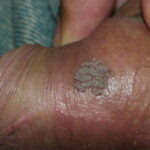
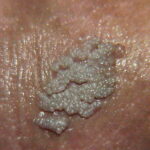
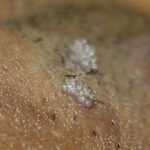
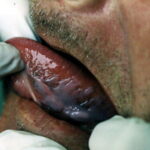
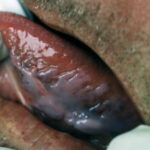
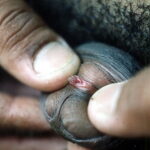
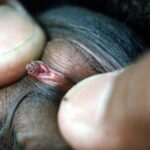
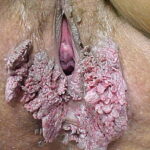
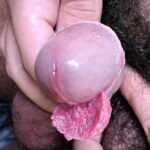
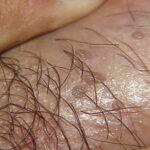
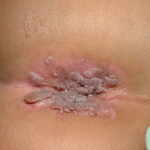
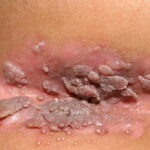
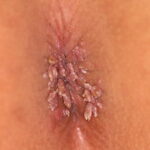
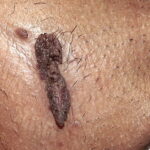
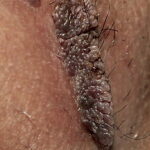
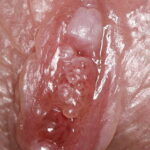
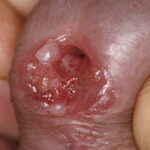
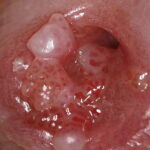
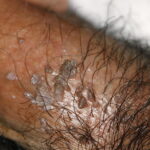
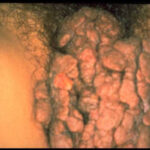
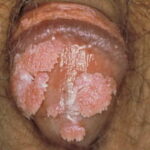
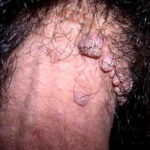
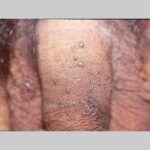
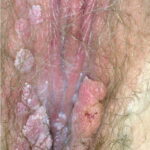
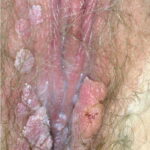
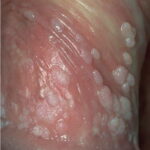
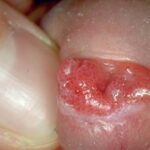
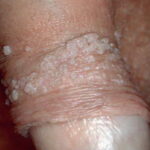
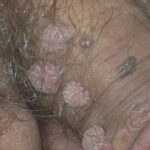
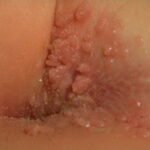
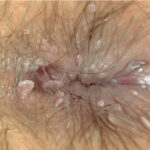
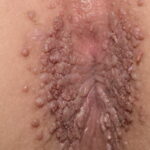
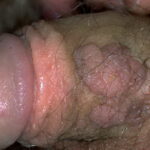
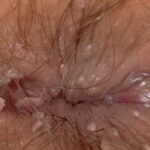
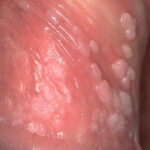
- Typical condyloma are usually discrete, papillary, cauliflowerlike lesions that involve multiple sites on moist surfaces.
- Keratotic warts are often seen on dry surfaces like the labia majora.
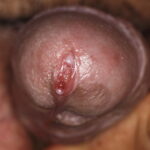
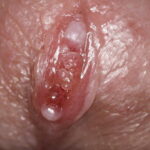
- Subclinical infection is another common presentation of condyloma. Tiny, slightly raised areas can be felt or visualized on the vagina or cervix.
- These flat warts are best visualized using 3-5% acetic acid and a colposcope. Areas infected with HPV appear acetowhite.
- Often, a biopsy is needed to distinguish these lesions from cervical squamous intraepithelial lesions or vaginal intraepithelial lesions.
- The sexual partner or partners of a woman with condyloma should be examined by a physician and treated if indicated. Often the examination of the male fails to reveal any visible condyloma.
Causes
Approximately 40 different types of HPV can infect the anogenital tract.
- Infection caused by the HPV virus results in local infections and appears as warty papillary condylomatous lesions.
- HPV infections in the genital area are sexually transmitted.
HPVs associated with genital tract lesions have been divided into low risk and high risk based on each genotype’s association with benign or malignant lesions. Most genital condylomata are due to infection by HPV-6 or HPV-11. These HPV types replicate as an episome and rarely incorporate their genetic material into the host DNA. In contrast, HPV-16 and HPV-18 can be recovered in approximately 70% of squamous cell carcinomas of the cervix. These high-risk HPV types, along with types 31, 33, 45, 51, 52, 56, 58, and 59 incorporate a portion of their genetic material into the host DNA. The E6 and E7 genes can produce oncoproteins that alter cell growth regulation. Specifically, E6 oncoprotein inactivates the tumor suppressor gene p53, and the oncoprotein produced by E7 inactivates pRB (retinoblastoma).
Laboratory Studies
Patients who present with condyloma acuminata do not necessarily need other laboratory studies. Patients who are diagnosed with condyloma are at an increased risk for other STDs.
- Consider testing for chlamydia, gonorrhea, syphilis, hepatitis B, hepatitis C, herpes, and HIV depending on the clinical situation.
- These patients need a Papanicolaou test of the cervix in accordance with the guidelines of the American College of Obstetricians and Gynecologists.
Histologic examination of the vulvar lesions to detect vulvar condyloma is sometimes difficult.
- Non-HPV conditions, such as vestibular papillomatosis and inflammatory squamous metaplasia, may be difficult to distinguish from condyloma with light microscopy.
- The pathologist may report microscopic features from a biopsy of the vulva that are suggestive, but not diagnostic, of HPV.
- When the histologic diagnosis of condyloma is questionable, HPV testing may be useful.
- A wide variety of methods to detect HPV have been used since 1983.
- Currently, the 2 most accurate methods use 2 consensus primer polymerase chain reaction (PCR) systems. The commercially available system is the Hybrid Capture II system with differential testing for 9 high-risk HPV types and 5 low-risk HPV types. No genotype-specific information is obtained from this test. Newer HPV tests that are being developed include DNA chip, Linear Array, and cycle sequencing.41
- Testing for HPV confirmation of equivocal vulvar histology results provides an objective method for confirming a diagnosis of condyloma.
Imaging Studies
No imaging studies are indicated.
Procedures
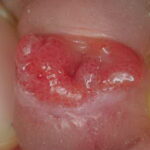
Patients who present with typical appearing condyloma acuminata usually do not need a vulvar biopsy. A biopsy is recommended for the following scenarios:
- Women with a history of vulvar dysplasia
- Postmenopausal women
- Women who fail medical therapy
- Clinical doubt about the diagnosis
Biopsy technique
- The base of the lesion is injected with 1% lidocaine.
- A biopsy can be performed easily with an alligator mouth biopsy forceps.
- Silver nitrate applied to the base of the biopsy site controls any bleeding.
- Rarely, a suture is required to obtain hemostasis.
Histologic Findings
Biopsy of the vulvar skin associated with condyloma shows evidence of hyperkeratosis, acanthosis, and parakeratosis. A chronic inflammatory infiltrate is often observed within the dermis. Koilocytosis, which is perinuclear cytoplasmic halos, is commonly observed in the superficial epithelial cells. Other microscopic findings include basilar hyperplasia with binucleated and multinucleated cells and enlarged parabasal cells with a foamy nuclear chromatin.
Medical Care
A variety of medical treatments are available to remove condyloma acuminata; no single treatment regimen is superior. Patients should be informed that genital warts resolve spontaneously in 20-30% of women within 3 months.12
- The treatment strategy is to eliminate as many of the visible lesions as possible until the host immune system can control viral replication.
- Treatment is reserved for patients with visible vulvar condyloma.
- The type of treatment is influenced by previous therapies, pregnancy status, sexual behavior, immune status, and the patient’s willingness to comply with therapy.
- Development of a therapeutic vaccine against HPV is currently being investigated. Two published trials demonstrated either a clinical complete response or a clinical partial response in women with vulvar or vaginal dysplasia.42,43
- Patients who are HIV positive or immunosuppressed due to immunosuppressive drugs usually require more than 1 treatment method. Often, the condyloma in these patients is refractory to therapy.
- Regardless of the mode of therapy chosen, recurrence rates are high for any patient with condyloma acuminata. This can result in a high level of frustration for the patient and the physician.
Medical therapy should be the first option for most patients. The authors’ prefer the following 4 options for patient-applied therapy.44
- Podofilox: Our first choice, in a nonpregnant patient, is Podofilox gel or solution. This agent is applied twice daily for 3 consecutive days and repeated for up to 4 weeks. Side effects are minimal.
- Imiquimod: This is our second choice in a nonpregnant woman with vulvar condyloma. Application is 3 times weekly at bedtime for up to 16 weeks. Imiquimod should be washed off 8 hours after application.45 Several randomized trials have demonstrated that application of imiquimod 5% cream can result in complete resolution of genital warts in up to 50% of patients. Recurrence rates range from 19-23% at 6 months.46 The primary side effect is erythema, itching, and burning. This drug is expensive and is often not a treatment option in many health insurance plans.
- Sinecatechins (Veregen): A third option is Veregen ointment for medical treatment of anogenital warts. Several randomized, double-blind, placebo-controlled trials demonstrated a 58% clearance of external genital warts. A trial of 502 male and female patients with anogenital warts were treated 3 times per week for a maximum of 16 weeks. Complete clearance of all baseline and newly occurring warts was obtained in 57% of patients treated with 15% sinecatechins ointment.47 The primary side effects were erythema, pruritus, and pain.48
- 5-fluorouracil: Another option is 5-fluorouracil (Efudex) cream. This agent is applied to genital warts to cause a chemical desquamation. A meta-analysis of 6 trials involving 645 women concluded that topical treatment with 5-fluorouracil has a therapeutic effect. The data were unclear on the risks and benefits and further studies were recommended.49 Protection of the normal surrounding skin is imperative to prevent pain, burning, and ulceration. This therapy is often not tolerated by patients. Use of this agent should be limited.
Other options include the following:
- Podophyllin: Podophyllin is applied directly to warts but no more than 0.5 mL should be used with one treatment. Systemic absorption, ulceration, and pain are potential side effects of this therapy. Podophyllin is contraindicated in pregnancy. The authors of this article prefer Podofilox to podophyllin. Podofilox results in less toxicity and can be self-administered by the patient.
- Interferon: Intralesional injection of interferon has been used to treat condyloma. Locally-injected interferon appears to be more effective than systemic injection of interferon. A meta-analysis of 7 randomized controlled trials comparing interferon and placebo for the treatment of genital warts reported complete response rate of 45% and 16%, respectively. Recurrence rates were 21% for interferon and 34% for patients in the placebo group.50 Side effects including flu-like symptoms, fatigue, and pain. Interferon is contraindicated in pregnancy.
- Trichloroacetic acid (TCA): TCA, using 80-90% concentration, is the author’s first choice to treat vulvar/vaginal condyloma in pregnant women. TCA should be applied to the condyloma after pretreatment of the surrounding normal skin with petroleum jelly. This treatment usually requires weekly application for 4-6 weeks. The principle side effect is pain and burning if the TCA comes in contact with the normal skin.
Surgical Care
Surgical removal of warts is appropriate if the condyloma do not respond to medical therapy, if there are numerous, bulky condyloma, or if the condyloma are associated with vulvar dysplasia. Several options are available.
- Simple excision
- Simple excision is usually performed in an outpatient surgical suite.
- The individual lesions are removed with a knife after general or regional anesthesia is administered.
- This procedure is reserved for refractory cases or extensive disease. The surgical specimen should be submitted for microscopic analysis.
- Reports in the literature indicate that within one year of surgery, complete wart clearance occurs in 35-72% of individuals treated with surgical excision. One report found surgical excision as effective as laser surgery.51
- Patients with a few small lesions can have vulvar condyloma removed in the office. The underlying skin should be anesthetized with 1% Xylocaine and the condyloma removed with a #15 knife blade. One or 2 sutures may be needed to reapproximate the healthy skin.
- Carbon-dioxide laser therapy51,52,53
- Laser treatment of vulvar condyloma acuminata effectively destroys the condyloma while sparing adjacent healthy tissue.
- This procedure is performed in outpatient surgery with general or regional anesthesia.
- The amount of energy needed to remove a condylomatous lesion with the laser depends on parameters controlled by the surgeon. These parameters include the amount of wattage, the length of time the beam is fired, and the spot size on the tissue. Some researchers calculate the power density, which equals the power (watts)/area (cm2). No exact power density is needed to remove vulvar or vaginal condyloma. The surgeon needs to be flexible in the application of the laser for each patient. If the laser is calibrated to 20 watts, continuous mode, the spot size can be adjusted easily to provide the proper power density.54
- Most patients experience significant discomfort beginning 24 hours after surgery and require narcotic analgesia.
- Laser therapy should be reserved for recalcitrant cases of condyloma or extensive disease.
- Complete wart clearance after laser surgery has been reported to occur in 23-52% of patients within 3 years of surgery.
- The recurrence rates are similar to surgical excision.
- Electrosurgery55
- For isolated lesions unresponsive to topical therapy, electrosurgical techniques can be performed in the office with local anesthesia.
- The most popular method is to use a loop electrode that removes the lesion or lesions.
- Pain after surgery is common and can be treated with narcotic analgesics. Topical analgesics, such as lidocaine jelly, can be beneficial to some patients.
- A recurrence rate in one trial was 22% compared with 44% for podophyllin resin.
- Cryotherapy56,57
- Cryotherapy is our treatment of choice for pregnant women when TCA fails to eliminate vulvar condyloma. This therapy is most effective for small lesions that can be treated with a small cryoprobe.
- Data from several clinical trials report a 63-88% clearance 3 months after therapy. A trial comparing cryotherapy to cryotherapy combined with podophyllotoxin failed to demonstrate an improved outcome with the combination therapy.58
- The recurrence rate of 22% is similar to electrosurgery.
- The primary drawbacks are discomfort, ulceration, or scabbing at the treatment site.
Activity
- The patient should refrain from sexual contact after any surgical procedure for condyloma acuminata.
- Soaking the genital area in warm water or sitz baths usually offers excellent pain relief.
- The genital area should be dried gently with a towel or a hair dryer.
- Loose fitting cotton underwear is helpful to avoid chafing.
- No other activity restrictions exist, although patients often have trouble sitting for long periods of time in the first week after surgery.
- Patients who have condyloma removed from the periurethral area may experience dysuria. Sitz baths and topical analgesics are beneficial.
No one curative treatment exists for condyloma acuminata.59 Simple topical therapies are the initial treatments of choice for most patients. They are cost effective and result in minimal toxicities. Most result in a 30-90% success rate in eliminating visible condyloma. Many clinical studies using topical therapies are not well designed, making comparisons between therapies difficult.
Antimitotics
Arrests dividing cells in mitosis, resulting in death of proliferating cells.
Podophyllum resin (Podocon-25, Podofin)
Treatment results in necrosis of visible wart tissue. Exact mechanism of action is unknown. Great variability exists in the potency of podophyllin between batches. American podophyllum contains one-fourth the amount of the Indian source. Warts visible after 6 treatments usually do not respond to further therapy.60
- Dosing
- Interactions
- Contraindications
- Precautions
Adult
Apply concentration of 25% sparingly onto lesions; wash treatment area 4 h after application; repeat q1-2wk until eliminated
Pediatric
Apply as in adults
- Dosing
- Interactions
- Contraindications
- Precautions
None reported
- Dosing
- Interactions
- Contraindications
- Precautions
Documented hypersensitivity; diabetes; impaired peripheral circulation; avoid use on mucous membranes, eyes, bleeding warts, moles, birthmarks, or unusual warts with hair
- Dosing
- Interactions
- Contraindications
- Precautions
Pregnancy
X – Contraindicated; benefit does not outweigh risk
Precautions
Powerful caustic and severe irritant; do not use if surrounding tissue is swollen or irritated; do not use large amounts; avoid contact with cornea; should be applied by a physician or trained nurse; redness or burning of the skin can occur 6-24 h after treatment
Podofilox (Condylox)
Topical antimitotic that can be synthesized chemically or purified from plant families Coniferae and Berberidaceae (eg, Juniperus and Podophyllum species).
Active agent of podophyllin resin and is available as a 0.5% solution. Can apply solution to warts at home.
- Dosing
- Interactions
- Contraindications
- Precautions
Adult
Apply 0.5% solution to warts bid for 3 d; repeat qwk for up to 4 wk
Pediatric
Apply as in adults
- Dosing
- Interactions
- Contraindications
- Precautions
None reported
- Dosing
- Interactions
- Contraindications
- Precautions
Documented hypersensitivity
- Dosing
- Interactions
- Contraindications
- Precautions
Pregnancy
C – Fetal risk revealed in studies in animals but not established or not studied in humans; may use if benefits outweigh risk to fetus
Precautions
Avoid contact with eyes; if eye contact occurs, immediately flush eye with copious quantities of water and seek medical advice; not for use on mucous membranes of genital area, including urethra, rectum, and vagina; do not exceed frequency of application or duration of usage
Antineoplastic agents
Topical preparation containing the fluorinated pyrimidine, 5-fluorouracil. Antineoplastic and antimetabolite agent.
Fluorouracil topical (Efudex)
Interferes with DNA synthesis by blocking methylation of deoxyuridylic acid, inhibiting thymidylate synthetase and, subsequently, cell proliferation. Limited data exist concerning the efficacy of this therapy for genital warts. Three case series indicate wart clearance in 10-50% of participants.61 Experimental treatments injecting 5-FU with epinephrine and bovine collagen currently are in trials.
- Dosing
- Interactions
- Contraindications
- Precautions
Adult
Apply 5% solution to warts 1-3 times per wk; wash off after 8 h
Pediatric
Not established
- Dosing
- Interactions
- Contraindications
- Precautions
None reported
- Dosing
- Interactions
- Contraindications
- Precautions
Documented hypersensitivity; potentially serious infections
- Dosing
- Interactions
- Contraindications
- Precautions
Pregnancy
X – Contraindicated; benefit does not outweigh risk
Precautions
Incidence of inflammatory reactions may occur with occlusive dressings; porous gauze dressing may be applied for cosmetic reasons without increase in reaction; adjacent healthy skin around warts should be coated with a protective gel before application; reproductive age group should use adequate contraception during therapy
Desiccants
These are acids that are most effective when applied to moist warts. They are nontoxic and can be used in pregnancy.
Trichloroacetic acid (Tri-Chlor)
Cauterizes skin, keratin, and other tissues. Although caustic, causes less local irritation and systemic toxicity than others in the same class; however, response often is incomplete and recurrence occurs frequently.62
- Dosing
- Interactions
- Contraindications
- Precautions
Adult
Apply 50-85% solution to warts q1-2wk in physician’s office; wash off after 4-6 h
Pediatric
Administer as in adults
- Dosing
- Interactions
- Contraindications
- Precautions
None reported
- Dosing
- Interactions
- Contraindications
- Precautions
Documented hypersensitivity; not for use on premalignant or malignant lesions
- Dosing
- Interactions
- Contraindications
- Precautions
Pregnancy
C – Fetal risk revealed in studies in animals but not established or not studied in humans; may use if benefits outweigh risk to fetus
Precautions
External use only; restrict use to treatment areas only; skin adjacent to warts needs to be protected; severe burning may occur
Immune response modifiers
Stimulates production of cytokines and has demonstrated strong antiviral activity.
Imiquimod (Aldara)
Induces secretion of interferon alpha and other cytokines. Mechanism of action unknown.63
- Dosing
- Interactions
- Contraindications
- Precautions
Adult
Apply 5% cream 3 times per wk hs; leave on skin for 6-10 h; treatment period not to exceed 16 wk
Pediatric
Administer as in adults
- Dosing
- Interactions
- Contraindications
- Precautions
None reported
- Dosing
- Interactions
- Contraindications
- Precautions
Documented hypersensitivity
- Dosing
- Interactions
- Contraindications
- Precautions
Pregnancy
B – Fetal risk not confirmed in studies in humans but has been shown in some studies in animals
Precautions
Not recommended for treatment of rectal, cervical, intravaginal, urethral, and intra-anal human papilloma infection; following surgery or drug treatment, do not use topical imiquimod until genital/perianal tissue is healed; local skin erythema, erosion, or abrasion can occur
Interferon alfa 2b (Intron)
Interferons have been used in the United States for the treatment of genital warts in various doses and preparations. Topical, intralesional, and systemic therapy have been used. Currently, no convincing evidence suggests that topical or systemic therapy is better than placebo.64,65,4,66
- Dosing
- Interactions
- Contraindications
- Precautions
Adult
1 million U per lesion administered directly into the wart 3 times per wk for 3 wk; no more than 5 warts should be treated at once
Pediatric
Administer as in adults
- Dosing
- Interactions
- Contraindications
- Precautions
Theophylline may increase toxicity; cimetidine may increase antitumor effects; zidovudine and vinblastine may increase toxicity
- Dosing
- Interactions
- Contraindications
- Precautions
Documented hypersensitivity
- Dosing
- Interactions
- Contraindications
- Precautions
Pregnancy
C – Fetal risk revealed in studies in animals but not established or not studied in humans; may use if benefits outweigh risk to fetus
Precautions
Depression and suicidal ideation may be adverse effects of treatment; flulike symptoms (eg, fever, dizziness, malaise, myalgia, headache) may occur
Vaccines
A human papillomavirus (HPV) quadrivalent vaccine is now available for prevention of HPV-associated dysplasias and neoplasias, including cervical cancer, genital warts (condyloma acuminata), and precancerous genital lesions. The immunization series should be completed in boys and girls, and young men and women aged 9-26 years.
Papillomavirus vaccine (Gardasil)
Quadrivalent HPV recombinant vaccine.
First vaccine indicated to prevent cervical cancer, genital warts (condyloma acuminata), and precancerous genital lesions (eg, cervical adenocarcinoma in situ; cervical intraepithelial neoplasia grades 1, 2, and 3; vulvar intraepithelial neoplasia grades 2 and 3; vaginal intraepithelial neoplasia grades 2 and 3) due to HPV types 6, 11, 16, and 18. Vaccine efficacy mediated by humoral immune responses following immunization series. Indicated for prevention of condyloma acuminata caused by HPV types 6 and 11 in boys, men, girls, and women aged 9-26 years.
- Dosing
- Interactions
- Contraindications
- Precautions
Adult
<26 years: 0.5 mL IM administered as 3 separate doses; administer second and third doses 2 and 6 mo after first dose, respectively
>26 years: Not established
Pediatric
<9 years: Not established
>9 years: Administer as in adults
- Dosing
- Interactions
- Contraindications
- Precautions
Immunosuppressive therapies (eg, irradiation, antineoplastic agents, corticosteroids) may decrease immune response to vaccine
- Dosing
- Interactions
- Contraindications
- Precautions
Documented hypersensitivity
- Dosing
- Interactions
- Contraindications
- Precautions
Pregnancy
B – Fetal risk not confirmed in studies in humans but has been shown in some studies in animals
Precautions
Shake well before administering; administer in deltoid region of upper arm or in higher anterolateral thigh; individuals with impaired immune responsiveness (eg, HIV infection, neoplastic disease, currently taking immunosuppressive drugs) may not elicit antibody response; because of IM administration, do not administer to individuals with bleeding disorders (eg, thrombocytopenia, coagulation disorders, anticoagulant therapy); common adverse effects include pain, swelling, erythema, and/or pruritus at injection site and fever
Miscellaneous topical ointment
Another topical product that has gained FDA approval for genital warts is kunecatechins.
Kunecatechins (Veregen)
Botanical topical drug product consisting of extract from green tea leaves. Mode of action unknown but does elicit antioxidant activity in vitro. Indicated for topical treatment of external genital and perianal warts (condylomata acuminatum) in immunocompetent patients.
- Dosing
- Interactions
- Contraindications
- Precautions
Adult
Apply topically tid; use approximately a 0.5-cm strand of ointment topically for each external genital or perianal wart