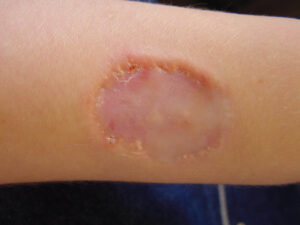
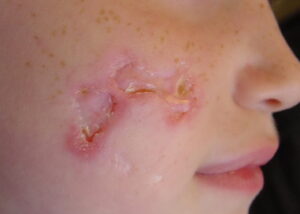
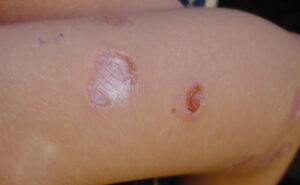
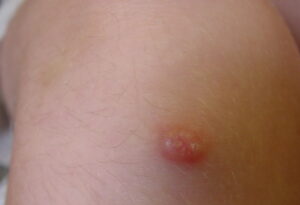
Churg strauss granuloma
Allergic granulomatosis and angiitis is a disorder characterized by extravascular granulomas, hypereosinophilia, and pulmonary and systemic small-vessel vasculitis. The combination of allergic granulomatosis and angiitis is associated with asthma, typically of adult onset, and allergic rhinitis.1 Churg and Strauss first described this disorder in 1951, when they reviewed 13 autopsy cases that were previously classified as polyarteritis nodosa. These cases were atypical in that asthma and an eosinophilia preceded the systemic vasculitis. They named the syndrome “allergic angiitis and allergic granulomatosis,” which has also come to be known as Churg-Strauss syndrome (CSS).2
Pathophysiology
The diagnosis of Churg-Strauss syndrome (allergic granulomatosis) is challenging because of the highly variable presentation and course of the disease. Some patients have only mild manifestations, while others are affected by life-threatening conditions. Some investigators have divided Churg-Strauss syndrome (allergic granulomatosis) into 3 phases, as follows3 :
- A prodromal phase characterized by allergic manifestations followed by asthma
- A second phase of marked peripheral blood eosinophilia and eosinophilic tissue infiltration that produces a picture similar to that of Loeffler syndrome, chronic eosinophilic pneumonia, or eosinophilic gastroenteritis
- Systemic vasculitis
Pulmonary involvement, neuropathy, and skin lesions are common with each occurring in at least two thirds or more of affected patients. Other systemic features include polyneuropathy (symmetric or mononeuritis multiplex), ischemic bowel disease, nasal perforation, glomerulonephritis, ocular inflammation, coronary arteritis, and cardiomyopathy.4 Myocardial involvement or congestive heart failure is the most common cause of death. An extremely high eosinophilia count is present in all patients, averaging 1 X 109/L, and approximately two thirds have a positive perinuclear antineutrophil cytoplasmic antibody (ANCA) titer, which targets primarily myeloperoxidase.5
More than one classification scheme exists for Churg-Strauss syndrome (allergic granulomatosis), including Lanham’s criteria, which emphasize clinical features, and the Chapel Hill Consensus Conference criteria, which emphasize pathology. A third option is the American College of Rheumatology (ACR) criteria, originally created for epidemiologic and therapeutic studies. The ACR diagnostic protocol requires the fulfillment of at least 4 of 6 criteria in the setting of vasculitis. These criteria are as follows6 :
- Asthma
- Eosinophilia count greater than 10% on a differential WBC count
- Mononeuropathy or polyneuropathy attributable to a vasculitis
- Transient pulmonary infiltrates
- Paranasal sinus abnormalities
- Biopsy specimen containing a blood vessel with extravascular eosinophils
The ACR criteria have a sensitivity of 85% and a specificity of 99.7%.7
History
The 3 phases—allergic, eosinophilic, and vasculitic—do not necessarily follow one another in any particular order. Symptoms depend on the phase and organ systems involved. A careful history should include medication usage, infectious symptoms, and/or preexisting disease.11
- Allergic phase of Churg-Strauss syndrome (allergic granulomatosis)
- Rhinitis, sinus pain, headache
- Cough
- Wheezing
- Eosinophilic phase of Churg-Strauss syndrome (allergic granulomatosis)
- General – Weight loss, fever, sweats
- Gastrointestinal – Abdominal pain, diarrhea, bleeding
- Pulmonary – Cough
- Vasculitic phase of Churg-Strauss syndrome (allergic granulomatosis)
- General – Malaise, lassitude, fever
- Cardiac – Chest pain, dyspnea
- Cutaneous – purpura, papules
- Pulmonary – Cough, hemoptysis
- Rheumatologic – Arthralgia, arthritis, myalgia
- Neurologic – Weakness, numbness
Physical
Clinical findings in Churg-Strauss syndrome (allergic granulomatosis) vary depending on the phase and organ systems involved.1
- Allergic phase Churg-Strauss syndrome (allergic granulomatosis)
- Nasal polyps
- Wheezing
- Cough
- Rhinitis
- Sinus tenderness
- Eosinophilic phase Churg-Strauss syndrome (allergic granulomatosis)
- General – Weight loss, fever, sweats
- Pulmonary – Cough, hemoptysis, rales, rhonchi
- Gastrointestinal – Rebound, masses, obstruction, ascites, bleeding
- Vasculitic phase Churg-Strauss syndrome (allergic granulomatosis)
- Constitutional symptoms – Fever, weight loss, adenopathy
- Cardiac – Gallop, pericardial friction rub, jugular venous distension, peripheral edema
- Pulmonary – Rales, rhonchi
- Nervous system – Mononeuritis multiplex, diffuse peripheral neuropathy (most often in a glove-and-stocking distribution), loss of a visual field, cerebral hemorrhage, infarction
- Renal – Mild proteinuria and hematuria
- Genitourinary – Obstructive uropathy
- Ocular – Episcleritis, panuveitis, marginal corneal ulceration, conjunctival infiltration, retinal infarction
- Musculoskeletal – Joint swelling, muscle tenderness
- Cutaneous
- Erythematous macules and papules at times resembling urticaria: Papules are most commonly located symmetrically on the extremities and scalp.
- Hemorrhagic lesions, including petechiae, extensive ecchymoses, and hemorrhagic bullae
- Subcutaneous nodules, facial edema, and livedo reticularis
Causes
The etiology of Churg-Strauss syndrome (allergic granulomatosis) remains unclear. Several triggers are suspected, including environmental factors and medications. Inhaled allergens, vaccinations, infections (both bacterial and parasitic), and prescription drugs (eg, carbamazepine, quinine, macrolides, corticosteroid-sparing drugs used to treat asthma) have all been implicated. A class of medications known as leukotriene inhibitors (montelukast, pranlukast, zafirlukast) has also been associated with Churg-Strauss syndrome (allergic granulomatosis). However, whether they are a direct cause or simply associated with the disease remains unclear at this time. Substantial tapering of corticosteroids with the use of leukotriene modifiers may allow unmasking of a previously unsuspected underlying disease, but some patients have never been on corticosteroids.12
Another possible drug association with Churg-Strauss syndrome (allergic granulomatosis) is omalizumab, an anti-immunoglobulin E (IgE) antibody used to treat asthma. Once again, whether omalizumab use is truly responsible for Churg-Strauss syndrome (allergic granulomatosis) or simply unmasks it after corticosteroid tapering remains unclear.13,14
A foreign or infectious agent has been suggested to initiate an inflammatory cascade in an individual with a susceptible genetic background. The inflammation then leads to eosinophilia. High levels of cytotoxic enzymes released by eosinophils have been found in the urine, sera, and bronchoalveolar lavage fluids of Churg-Strauss syndrome (allergic granulomatosis) patients. These enzymes could result in oxidative tissue damage.15 ANCA antibodies activate neutrophils in vitro, producing greater quantities of reactive oxygen species, which may result in endothelial cell damage. ANCA-positive patients typically have more prominent vasculitic manifestations.12 However, the etiology of Churg-Strauss syndrome (allergic granulomatosis) remains incompletely understood, particularly in ANCA-negative patients.Laboratory Studies
- CBC count with erythrocyte sedimentation rate (ESR): Eosinophilia greater than 10% is a diagnostic criterion of the American College of Rheumatology (ACR). Treatment with steroids may mask this finding. Anemia, leukocytosis, thrombocytosis, and an elevated ESR usually accompany the vasculitic phase of Churg-Strauss syndrome (allergic granulomatosis).
- Rheumatoid factor (RF) and antinuclear antibody (ANA) testing: Some patients may have weakly positive ANA and/or RF results.
- Antineutrophilic cytoplasmic antibodies (ANCA) testing: Perinuclear ANCA directed predominantly against a myeloperoxidase was initially reported in as many as 75-80% of patients with Churg-Strauss syndrome (CSS), but in one study showed that only 13% had positive perinuclear ANCA findings. Because of the low sensitivity of this test, a negative ANCA result does not rule out Churg-Strauss syndrome (allergic granulomatosis). Classic ANCA directed against serine proteinase 3 is positive in approximately 10% of patients with Churg-Strauss syndrome (allergic granulomatosis). High titers of classic ANCA are seen more often with Wegener granulomatosis than with Churg-Strauss syndrome (allergic granulomatosis).
- Systemic vasculitis testing: Screened patients suspected to have systemic vasculitis for cryoglobulins, antiphospholipid antibodies, immunoglobulins (immunoglobulins G, A, E, and M), complement levels (CH50, C3, and C4). Immunoglobulin E levels are usually elevated in persons with Churg-Strauss syndrome (allergic granulomatosis).
- Liver function testing: Serum aspartate aminotransferase and serum alanine aminotransferase levels may indicate liver or muscle involvement. Hepatitis B and C virus serologies are recommended for suspected cases of systemic vasculitis.
- Creatine phosphokinase testing: Creatine phosphokinase levels may indicate muscle or cardiac involvement.
- Gastrointestinal testing: Stool guaiac screening can be performed to test for bleeding.
- BUN and creatinine measurements: Kidney involvement in the form of focal segmental glomerulonephritis may occur, but it is typically mild. Severe kidney involvement is more common in the other systemic vasculitides, such as Wegener granulomatosis and polyarteritis nodosa, than in Churg-Strauss syndrome (allergic granulomatosis).
- Urinalysis: Hematuria and proteinuria have been reported but are usually mild. Severe kidney involvement is more typical of the other systemic vasculitides than of Churg-Strauss syndrome (allergic granulomatosis).1,7
Imaging Studies
- Chest radiography: Findings may be abnormal in both the eosinophilic stage and the vasculitic phase. Radiographs typically reveal transient patchy alveolar opacities, nodular infiltrates, or a diffuse interstitial pattern. Bilateral hilar adenopathy has also been reported.5
- Sinus radiography: Opacification of the paranasal sinuses is an ACR diagnostic criterion.
- ECG, echocardiography, and cardiac MRI: Cardiac involvement is common in Churg-Strauss syndrome (allergic granulomatosis) and may affect the pericardium, myocardium, and endocardium. Pericardial effusion, cardiomyopathy, cardiac valve abnormalities, coronary vasculitis, and granulomatous myocarditis have all been described.11
Other Tests
- Any additional testing should be based on clinical history and patient symptoms.
Procedures
- Tissue biopsy: Due to ease of access, the skin, followed by the kidney, are the most frequently selected sites for biopsy. Skin biopsy specimens should be taken from a lesion less than 48 hours old. Biopsy specimens for direct immunofluorescence examination should also be obtained from a lesion less than 48 hours old.
Histologic Findings
Biopsy may reveal a variety of histologic changes, including necrotizing vasculitis of small-to-medium vessels, eosinophilic infiltration, and extravascular granulomas. Nodular lesions frequently reveal characteristic extravascular granulomas. These granulomas are not pathognomic for Churg-Strauss syndrome (allergic granulomatosis) because they are also found in persons with Wegener granulomatosis, polyarteritis nodosa, systemic lupus erythematosus, rheumatoid arthritis, lymphoproliferative disorders, and other immunoreactive disorders. Granulomas are composed of a central core of necrotic eosinophilic debris with degeneration of collagen and surrounded by a peripheral palisade of epithelioid histiocytes with few, if any, giant cells. The non-nodular lesions most often demonstrate a vasculitis or a nonspecific perivascular infiltrate of eosinophils and mononuclear cells without vasculitis.
Medical Care
Churg-Strauss syndrome (CSS) is typically a corticosteroid-responsive disease; hence, they are first-line therapeutic agents. Most patients respond to corticosteroid monotherapy. Intravenous corticosteroids should be considered for patients with extensive disease. Dramatic response to therapy may be observed with improvement in eosinophilia counts, reduction of the erythrocyte sedimentation rate (ESR), and reduction in muscle enzyme levels within 1-2 weeks of initiation of treatment. Corticosteroids may be tapered once clinical improvement occurs. Importantly, note that patients with Churg-Strauss syndrome (allergic granulomatosis)–related neuropathy respond more slowly to treatment. Residual asthma or other symptoms may require the continuation of low-dose prednisone therapy.4
Cyclophosphamide should be initiated in severely ill patients who do not respond to initial therapy. Patients with life-threatening disease or those at risk of organ failure are potential candidates. The addition of cyclophosphamide appears to improve outcomes and reduces the incidence of relapses. For patients with systemic disease who are not at risk for major organ failure or death, methotrexate (MTX) may be given as a corticosteroid-sparing agent to reduce the cumulative dose. Other immunomodulatory medications include azathioprine (AZA) and mycophenolate mofetil, which may also be used as corticosteroid-sparing agents. AZA is best used for maintenance therapy rather than for induction of remission in refractory disease. Chlorambucil and plasma exchange have occasionally been used and are most effective when used in combination therapy.7
Agents that block tumor necrosis factor (TNF), such as infliximab and etanercept, have been used for a limited period in severe life-threatening cases. These agents, when combined with corticosteroids or other immunomodulatory agents, greatly increase the risk of infection due to immunosuppression. More data are needed to determine whether these drugs have a favorable risk-to-benefit ratio for use in Churg-Strauss syndrome (allergic granulomatosis) patients. Another infrequently used therapy is recombinant interferon (IFN) alfa, which can be effective when given on a short-term basis in otherwise refractory cases. One anecdotal case report has described the use of rituximab in a patient with recalcitrant Churg-Strauss syndrome (allergic granulomatosisMedication
Corticosteroids are the mainstay of treatment in Churg-Strauss syndrome (allergic granulomatosis). The addition of other medications may be necessary in cases of life- or organ-threatening vasculitis.
Corticosteroids
These drugs have anti-inflammatory and immunosuppressive properties.
Methylprednisolone (Medrol, Solu-Medrol)
To treat inflammatory and immune reactions. May decrease inflammation by reversing increased capillary permeability and suppressing PMN activity.
- Dosing
- Interactions
- Contraindications
- Precautions
Adult
15 mg/kg/d IV bolus, then convert to prednisolone
Pediatric
Administer as in adults
- Dosing
- Interactions
- Contraindications
- Precautions
Coadministration with digoxin may increase digitalis toxicity due to hypokalemia; estrogens may increase levels; phenobarbital, phenytoin, and rifampin may decrease levels (adjust dose); monitor for hypokalemia with concurrent diuretics; antacids decrease absorption; amphotericin B and carbonic anhydrase inhibitors may lead to hypokalemia
- Dosing
- Interactions
- Contraindications
- Precautions
Documented hypersensitivity; viral, fungal, or TB skin infections
- Dosing
- Interactions
- Contraindications
- Precautions
Pregnancy
C – Fetal risk revealed in studies in animals but not established or not studied in humans; may use if benefits outweigh risk to fetus
Precautions
Transient adverse effects of pulse IV corticosteroids are bitter taste, facial flushing, headache, asthenia, significant rise in blood pressure, and temporary glucose intolerance; consider restricting breastfeeding in patients taking >20 mg/d or withhold breastfeeding for 4 h after dosing; weight gain, Cushingoid appearance, osteoporosis, and avascular necrosis; use may increase risk of or worsen preexisting viral, fungal, opportunistic, and parasitic infections; may cause reactivate TB; increased risk of peptic ulcer disease, especially in patients with history; posterior subcapsular cataract formation; CNS complications (psychosis, agitation, insomnia, depression); may induce or worsen preexisting hypertension and glucose and lipid abnormalities; skin changes include atrophy, alopecia, acneiform eruptions, poor wound healing, purpura, striae, hirsutism, and desquamation
Prednisolone (Econopred, Articulose-50, Delta-Cortef)
Decreases autoimmune reactions, possibly by suppressing key components of immune system.
- Dosing
- Interactions
- Contraindications
- Precautions
Adult
20-60 mg PO qam or 1 mg/kg PO for 3 d
Pediatric
1-2 mg/kg PO qam
- Dosing
- Interactions
- Contraindications
- Precautions
Decreases effects of salicylates and toxoids (for immunizations); phenytoin, carbamazepine, barbiturates, and rifampin decrease effects of corticosteroids
- Dosing
- Interactions
- Contraindications
- Precautions
Documented hypersensitivity; viral, fungal or TB skin lesions
- Dosing
- Interactions
- Contraindications
- Precautions
Pregnancy
C – Fetal risk revealed in studies in animals but not established or not studied in humans; may use if benefits outweigh risk to fetus
Precautions
Caution in hyperthyroidism, osteoporosis, cirrhosis, nonspecific ulcerative colitis, peptic ulcer, diabetes, and myasthenia gravis
Immunomodulators
These agents modulate immune responses to various stimuli.
Methotrexate (Folex PFS, Rheumatrex)
Unknown mechanism of action in treatment of inflammatory reactions; may affect immune function. Ameliorates symptoms of inflammation (eg, pain, swelling, stiffness).
- Dosing
- Interactions
- Contraindications
- Precautions
Adult
7.5 mg/wk PO once or 2.5 mg bid for 3 doses qwk; adjust dose gradually for satisfactory response
Pediatric
5-15 mg/m2/wk PO/IM single dose or 3 divided doses q12h
- Dosing
- Interactions
- Contraindications
- Precautions
Oral aminoglycosides may decrease absorption and blood levels of concurrent oral MTX; charcoal lowers levels; coadministered etretinate may increase hepatotoxicity; folic acid or derivatives in some vitamins may decrease response; coadministration with NSAIDs may be fatal; indomethacin and phenylbutazone can increase plasma levels; may decrease phenytoin serum levels; probenecid, salicylates, procarbazine, and sulfonamides, including TMP-SMZ, may increase effects and toxicity; may increase plasma levels of thiopurines
- Dosing
- Interactions
- Contraindications
- Precautions
Documented hypersensitivity; alcoholism, hepatic insufficiency, immunodeficiency syndromes, preexisting blood dyscrasias (eg, bone marrow hypoplasia, leukopenia, thrombocytopenia, significant anemia)
- Dosing
- Interactions
- Contraindications
- Precautions
Pregnancy
X – Contraindicated; benefit does not outweigh risk
Precautions
Monitor CBC counts monthly and liver and renal function q1-3mo during therapy (more frequently during initial dosing, dose adjustments, or with risk of elevated MTX levels [eg, dehydration]); toxic effects on hematologic, renal, GI, pulmonary, and neurologic systems; discontinue if blood counts drop significantly; aspirin, NSAIDs, or low-dose steroids may be administered concomitantly (increased toxicity with NSAIDs or salicylates not tested)
Azathioprine (Imuran)
Antagonizes purine metabolism and inhibits synthesis of DNA, RNA, and proteins. May decrease proliferation of immune cells, lowering autoimmune activity.
- Dosing
- Interactions
- Contraindications
- Precautions
Adult
1 mg/kg/d PO for 6-8 wk; increase by 0.5 mg/kg q4wk until response or 2.5 mg/kg/d
Pediatric
Initial dose: 2-5 mg/kg/d PO/IV
Maintenance dose: 1-2 mg/kg/d PO/IV- Dosing
- Interactions
- Contraindications
- Precautions
Allopurinol increases toxicities; concurrent use with ACE inhibitors may induce severe leukopenia; may increase levels of MTX metabolites and decrease effects of anticoagulants, neuromuscular blockers, and cyclosporine
- Dosing
- Interactions
- Contraindications
- Precautions
Documented hypersensitivity; thiopurine methyltransferase deficiency
- Dosing
- Interactions
- Contraindications
- Precautions
Pregnancy
D – Fetal risk shown in humans; use only if benefits outweigh risk to fetus
Precautions
Increases risk of neoplasia; caution in liver disease and renal impairment; hematologic toxicities may occur; check thiopurine methyltransferase level before treatment; monitor CBC count and liver function periodically
Cyclosporine (Sandimmune, Neoral)
Cyclic polypeptide suppresses some humoral immunity and, to a greater extent, cell-mediated immune reactions (eg, delayed hypersensitivity, allograft rejection, experimental allergic encephalomyelitis, and graft vs host disease) in many organs. Base dosing on ideal body weight.
- Dosing
- Interactions
- Contraindications
- Precautions
Adult
Initial PO dose: 14-18 mg/kg/d 4-12 h before organ transplantation
Maintenance PO dose: 5-15 mg/kg/d qd or divided bid
Initial IV dose: 5-6 mg/kg qd 4-12 h before organ transplantation
Maintenance IV dose: 2-10 mg/kg/d divided q8-12hPediatric
Administer as in adults
- Dosing
- Interactions
- Contraindications
- Precautions
Carbamazepine, phenytoin, isoniazid, rifampin, and phenobarbital may decrease concentrations; azithromycin, itraconazole, nicardipine, ketoconazole, fluconazole, erythromycin, verapamil, grapefruit juice, diltiazem, aminoglycosides, acyclovir, amphotericin B, and clarithromycin may increase toxicity; acute renal failure, rhabdomyolysis, myositis, and myalgias increase with concurrent lovastatin
- Dosing
- Interactions
- Contraindications
- Precautions
Documented hypersensitivity; uncontrolled hypertension or malignancies; PUVA or UVB radiation in psoriasis (may increase risk of cancer)
- Dosing
- Interactions
- Contraindications
- Precautions
Pregnancy
C – Fetal risk revealed in studies in animals but not established or not studied in humans; may use if benefits outweigh risk to fetus
Precautions
Evaluate renal and liver functions often by measuring BUN, serum creatinine, serum bilirubin, and liver enzymes; may increase risk of infection and lymphoma; reserve IV for those who cannot take PO
Interferon alfa 2a and 2b (Roferon-A [alpha-2a], Intron A [alpha-2b])
Recombinant DNA product. Mechanism of antitumor activity not clearly understood; direct antiproliferative effects against malignant cells and modulation of host immune response may be important.
- Dosing
- Interactions
- Contraindications
- Precautions
Adult
2 million U/m2 SC 3 times/wk for 30 d
Pediatric
Not established
- Dosing
- Interactions
- Contraindications
- Precautions
Theophylline may increase toxicity; cimetidine may increase antitumor effects; zidovudine and vinblastine may increase toxicity, interleukin 2
- Dosing
- Interactions
- Contraindications
- Precautions
Documented hypersensitivity
- Dosing
- Interactions
- Contraindications
- Precautions
Pregnancy
C – Fetal risk revealed in studies in animals but not established or not studied in humans; may use if benefits outweigh risk to fetus
Precautions
Caution in brain metastases, severe hepatic or renal insufficiency, seizure disorders, multiple sclerosis, or compromised CNS
Alkylating agents
These agents are recommended as initial therapy of severe, life-threatening Churg-Strauss syndrome (allergic granulomatosis) and for patients who are not responsive to corticosteroids alone.
Cyclophosphamide (Cytoxan, Neosar)
Chemically related to nitrogen mustards. As an alkylating agent, mechanism of action of the active metabolites may involve cross-linking of DNA, which may interfere with growth of normal and neoplastic cells.
- Dosing
- Interactions
- Contraindications
- Precautions
Adult
0.6 g/m2 to 2.0 mg/kg/d infused over 1 h, followed by vigorous IV hydration; repeat 1 time/mo for 1 y; first pulse given on day 4 after third day of bolus methylprednisolone; sodium 2-mercaptoethane sulfonate (mesna) at 160% of cyclophosphamide dose separated into 4 doses at 0, 3, 6, and 9 h optional; may help prevent serious urologic adverse effects
Pediatric
Not recommended
- Dosing
- Interactions
- Contraindications
- Precautions
Allopurinol may increase risk of bleeding or infection and enhance myelosuppressive effects; may potentiate doxorubicin-induced cardiotoxicity; may reduce digoxin serum levels and antimicrobial effects of quinolones; chloramphenicol may increase half-life while decreasing metabolite concentrations; may increase effect of anticoagulants; coadministration with high doses of phenobarbital may increase metabolism and leukopenic activity; thiazide diuretics may prolong cyclophosphamide-induced leukopenia and neuromuscular blockade by inhibiting cholinesterase activity
- Dosing
- Interactions
- Contraindications
- Precautions
Documented hypersensitivity; severely depressed bone marrow function
- Dosing
- Interactions
- Contraindications
- Precautions
Pregnancy
D – Fetal risk shown in humans; use only if benefits outweigh risk to fetus
Precautions
Hematologic myelosuppression, primarily leukopenia, is most common adverse effect; nadir usually in 8-12 d; thrombocytopenia and anemia less frequent; GI adverse effects include anorexia, nausea, and emesis; PO ondansetron and dexamethasone may control nausea resistant to antiemetics; urologic adverse effects are dysuria, urgency, hematuria, bladder fibrosis, and necrosis; death from hemorrhagic cystitis has occurred (mesna may prevent); bladder cancer risk increased 45-fold; reproductive toxicities are azoospermia and amenorrhea; other adverse effects are hair loss, mucositis, and hyperpigmentation
Chlorambucil (Leukeran)
Alkylates and cross-links strands of DNA, inhibiting DNA replication and RNA transcription.
- Dosing
- Interactions
- Contraindications
- Precautions
Adult
0.1-0.2 mg/kg/d PO or 3-6 mg/m2/d for 3-6 wk; adjust dose depending on blood counts
Pediatric
0.1-0.2 mg/kg/d PO qd for 5-15 wk
- Dosing
- Interactions
- Contraindications
- Precautions
None reported
- Dosing
- Interactions
- Contraindications
- Precautions
Documented hypersensitivity; previous resistance to this medication
- Dosing
- Interactions
- Contraindications
- Precautions
Pregnancy
D – Fetal risk shown in humans; use only if benefits outweigh risk to fetus
Precautions
Caution in seizure disorders and bone marrow suppression
Anti-TNF agents
These agents are recommended in severe cases when corticosteroids and cyclophosphamide may be insufficient to induce remission.
Infliximab (Remicade)
Monoclonal antibody with human constant and murine variable regions; neutralizes biologic activity of TNF-alpha with high binding affinity to the soluble transmembrane forms of TNF-alpha and inhibits binding of TNF with receptors.
- Dosing
- Interactions
- Contraindications
- Precautions
Adult
3-20 mg/kg IV; repeat infusions q2-6wk
Pediatric
Not established
- Dosing
- Interactions
- Contraindications
- Precautions
Anakinra
- Dosing
- Interactions
- Contraindications
- Precautions
Active infection, moderate-to-severe CHF, hypersensitivity to murine products or other components
- Dosing
- Interactions
- Contraindications
- Precautions
Pregnancy
B – Fetal risk not confirmed in studies in humans but has been shown in some studies in animals
Precautions
Severe infection (including sepsis) reported, especially with concomitant immunosuppressive infections; histoplasmosis, coccidioidomycosis, listeriosis, Pneumocystis TB, other bacterial and fungal infections reported; in patients in areas with endemic histoplasmosis or coccidioidomycosis, carefully weigh benefits and risks
Etanercept (Enbrel)
Fusion protein of TNF receptor and Fc portion of human IgG-1; binds TNF and blocks interaction of TNF-alpha and TNF-beta with cell-surface receptors, rendering TNF biologically inactive.
- Dosing
- Interactions
- Contraindications
- Precautions
Adult
25-50 mg SC 2 times/wk
Pediatric
0.4 mg/kg/wk SC; not to exceed 25 mg
- Dosing
- Interactions
- Contraindications
- Precautions
Concomitant use of anakinra increases infection risk
- Dosing
- Interactions
- Contraindications
- Precautions
Active infection, immunization with live vaccines
- Dosing
- Interactions
- Contraindications
- Precautions
Pregnancy
B – Fetal risk not confirmed in studies in humans but has been shown in some studies in animals
Precautions
Allergic reactions reported; may worsen or increase infections; associated with life-threatening infectious disease besides TB (eg, candidiasis, histoplasmosis, aspergillosis, listeriosis); TNF-alpha antagonists often used with other immunosuppression, particularly glucocorticoids and MTX; increased rates of TB or other infections due to interactions among therapies unknown