Candidiasis
The Candida fungus is both normal flora and an invasive pathogen. The range of infection with Candida species varies from a benign local mucosal membrane infection to disseminated disease. Severe disease is typically associated with an immunocompromised state including those vulnerable to iatrogenic pathogens in the intensive care unit or those with predisposing immunologic conditions such as malignancy, organ dysfunction, or immunosuppressive therapy.
Candida is a unicellular yeast whose cells reproduce by budding. This organism can flourish in most environments. It frequently colonizes the oropharynx, skin, mucous membranes, lower respiratory, and gastrointestinal and genitourinary tracts. Pathogenesis occurs with increased fungal burden and colonization, such as in the setting of broad-spectrum antimicrobial agents; breakdown of normal mucosal and skin barriers, which can occur with indwelling intravascular devices, recent surgery/trauma or tissue damage secondary to chemotherapy or radiation; or immune dysfunction secondary to disease states or iatrogenic conditions.
Candidiasis is the most common opportunistic fungal infection. Disease manifestation of candidal infection can vary with type of host immunodeficiency. Lymphocytes and cell-mediated immunity are important in the prevention of mucosal candidiasis. Therefore, patients with T-cell deficiency, such as human immunodeficiency virus (HIV), have a propensity to develop recurrent and/or persistent mucocutaneous candidiasis. Patients with neutropenia are at risk for invasive candidiasis and candidemia as functioning monocytes and polymorphonuclear cells are responsible for killing pseudohyphae and blastospores. Complement and immunoglobulins are necessary for intracellular killing of the organisms and patients with deficiencies can have a more prolonged and complicated course
History
Candidal infection has a wide range of clinical manifestations from self-limited local mucocutaneous disease to severe sepsis with multiorgan system failure.
Local mucous membrane infections
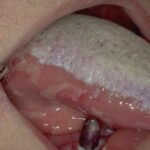
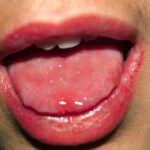
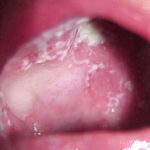
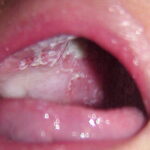
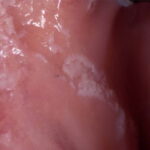
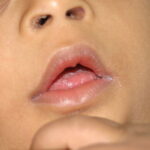
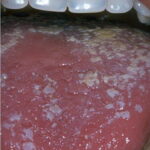
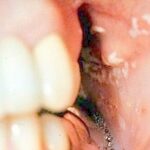
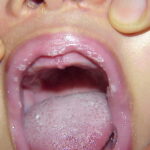
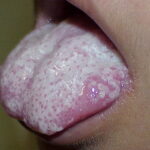
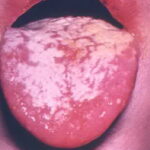
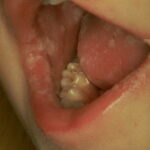
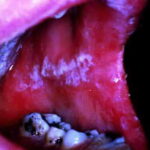
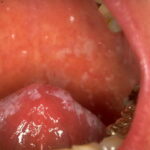
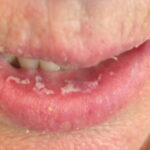
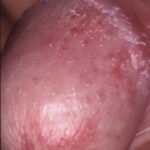
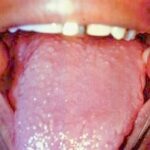
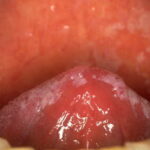
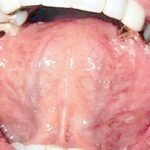
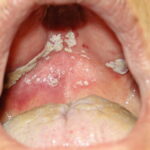
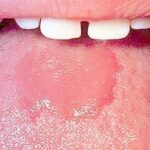
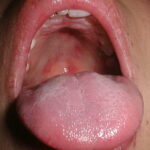
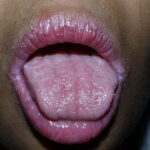
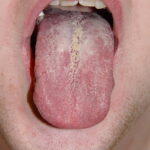
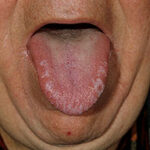
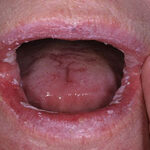
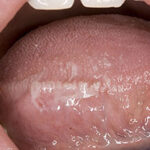
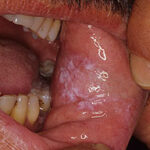
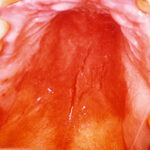
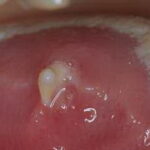
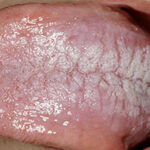
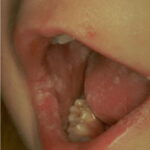
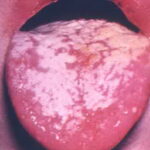
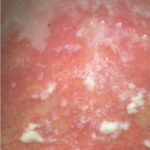
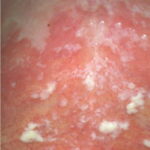
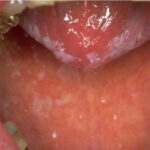
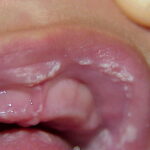
Oral candidiasis, also referred to as thrush, is characterized by creamy white, curd-like patches on the tongue and oral mucosa. These patches are a pseudomembrane of Candida, desquamated epithelial cells, leukocytes, bacteria, keratin, necrotic tissue, and food debris. Chronic atrophic candidiasis, or denture sore mouth, is a chronic inflammatory reaction with epithelial thinning under dental plates. Saliva usually protects oral mucosa against Candida and, in turn, dry mouth increases yeast counts. Dentures also create a reservoir of Candida incorporation into the biofilm covering the material and increases the risk factor for invasive candidosis, especially if the patient is immunocompromised.
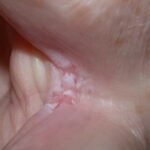
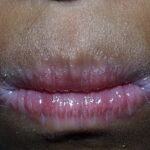
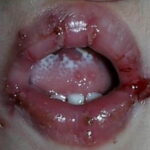
Candidal leukoplakia consists of firm, white plaques affecting the cheeks, lips, and tongue, which frequently have a protracted course and can be precancerous. Angular cheilitis is characterized as erythema and fissuring at the corners of the mouth.
Risk factors for oral infection include antibiotic use, immunodeficiency (up to 90% of HIV-positive persons will have a symptomatic episode of oropharyngeal candidiasis sometime during progression to AIDS), xerostomia, inhaled corticosteroids, and denture use. Symptoms include dry mouth, loss of taste, and occasionally, pain with eating.
Candida epiglottitis is another entity that can manifest for immunocompromised patients. It may exist as a coinfection with bacterial organisms or on its own. A KOH prep and fungal cultures may be helpful in treating epiglottitis that is not improving on antibiotics alone.7
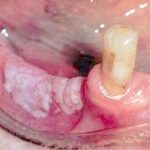
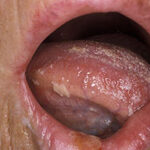
Candida esophagitis is most commonly associated with treatment of hematopoietic or lymphatic malignancies. It is also an AIDS-defining illness, though patients with Candida esophagitis and no underlying illness have been reported. Symptoms include dysphagia, sensation of obstruction on swallowing, retrosternal chest pain, nausea, and vomiting. Definitive diagnosis is made by endoscopic biopsy. On endoscopy, white plaques similar to thrush may be present; ulcerations, pseudomembrane formation, and diverticula may also be seen. Candidal esophagitis can occur concomitantly with herpes simplex and cytomegalovirus infection in severely immunocompromised patients.
Other locations of the gastrointestinal tract are often infected with Candida. Lesions may be found in the stomach and small and large intestines most commonly in patients with neoplastic disease. Candidal cholecystitis was found in a patient without any history of underlying malignancy.8
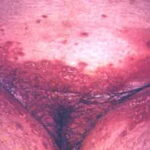
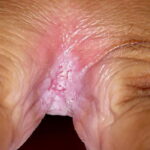
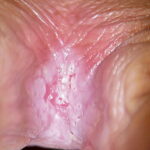
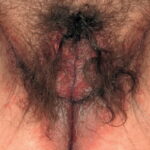
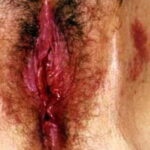
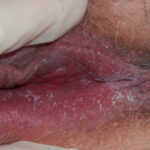
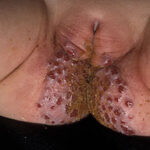
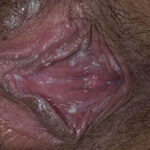
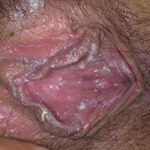
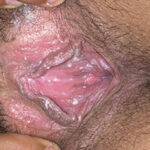
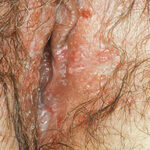
Candidal vaginitis is the most common form of mucosal candidiasis. Vulvovaginal candidiasis is usually secondary to overgrowth of normal flora Candida species in the vagina. Bacteria such as Lactobacillus acidophilus balance Candida and prevent yeast overgrowth and pathogenic infection. Conditions that disrupt the balance of normal vaginal flora include antibiotic use, oral contraceptives, contraceptive devices, high estrogen levels, and immunocompromised states such as diabetes mellitus and HIV. Another risk factor for vulvovaginal candidiasis may be intrauterine contraceptive devices.9
Of vulvovaginal candidiasis cases, 70-90% are caused by Candida albicans, while the remainder of infections are caused by C glabrata and C tropicalis. Symptoms classically are described as pruritus, vaginal irritation, and dysuria. Thick, curd-like discharge is often present, but scant discharge may also characterize infection. Vaginal edema and erythema are present on examination.
Epidemiologically, vaginal Candida infections are important as they may increase viral shedding in HIV-infected women.
Cutaneous candidiasis syndromes include the following:
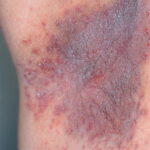
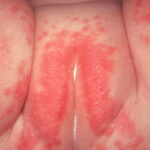
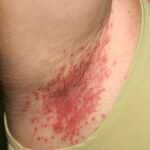
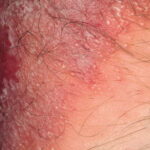
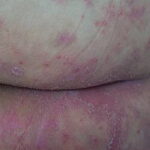
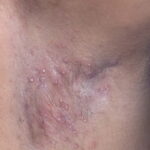
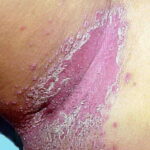
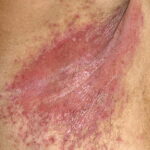
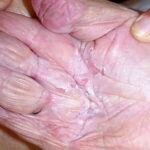
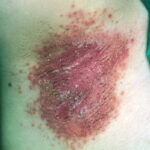
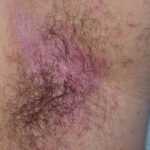
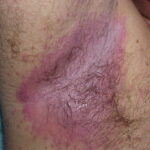
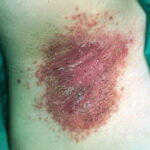
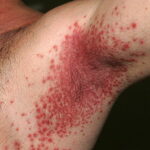
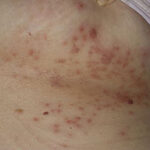
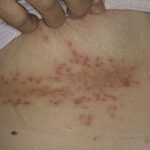
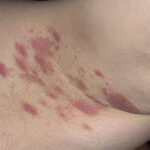
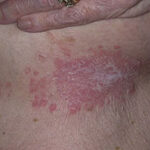
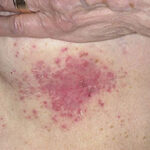
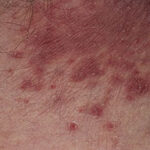
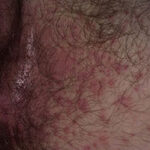
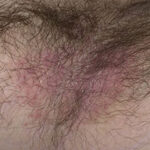
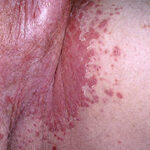
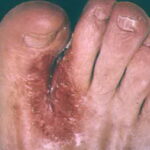
- Generalized cutaneous candidiasis: This infection is characterized by widespread eruptions with increased severity in the genitocrural folds, anal region, axillae, and hands and feet that occurs in both children and adults.
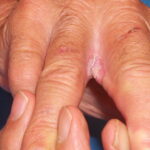
- Erosio interdigitalis blastomycetica: This is an intertriginous infection, which is predisposed to maceration.
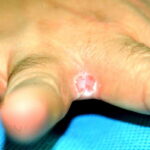
- Candida folliculitis: This is most frequently seen in immunocompromised hosts and among intravenous drug users.
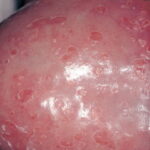
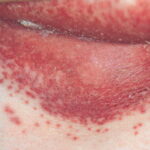
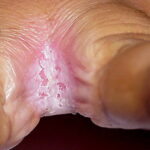
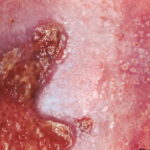
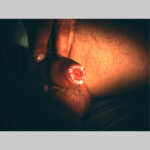
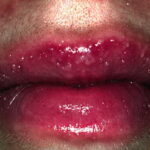
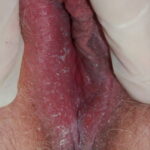
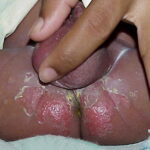
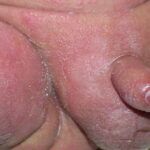
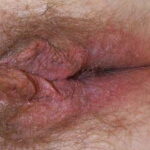
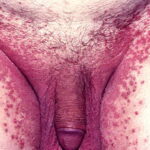
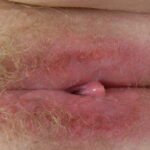
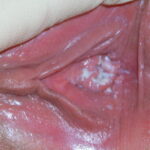
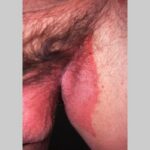
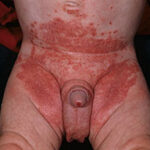
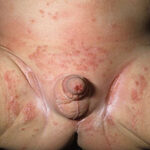
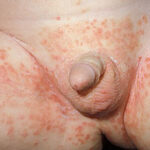
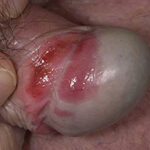
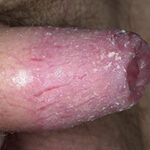
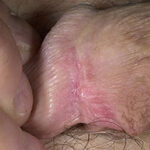
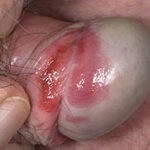
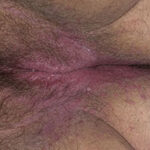
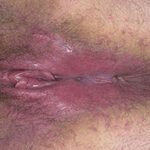
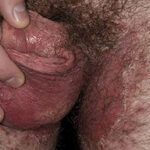
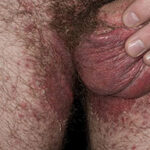
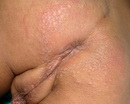
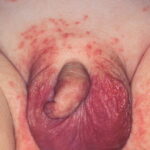
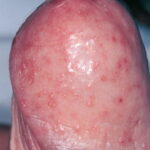
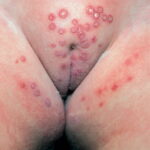
- Candida balanitis: This infection is usually acquired with sexual intercourse with an infected partner. Rash typically begins as vesicles on the penis that develop into patches similar in appearance to thrush (shown in the image below). Extension may occur to the scrotum and buttocks. Symptoms include burning and pruritus. C glabrata has been implicated as the cause of Fournier’s gangrene in an immunocompromised patient.10
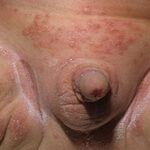
- Mammary candidiasis: This is a condition that can affect breastfeeding women.11
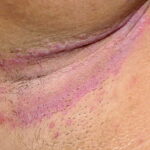
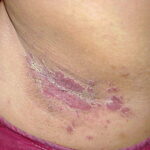
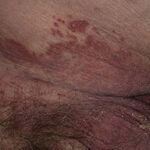
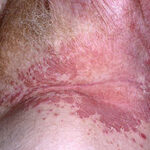
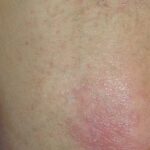
- Intertrigo: This develops in sites where skin surfaces are close in proximity. Lesions begin as vesicopustules that enlarge, rupture, and develop maceration and fissuring. Satellite lesions may be present. A variant form of cutaneous candidiasis in the intertriginous region has a miliary appearance.
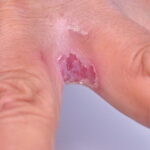
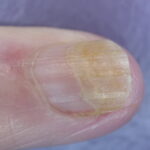
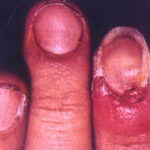
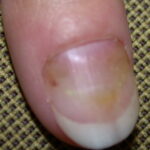
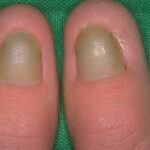
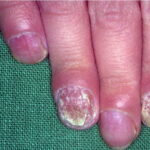
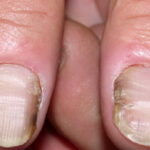
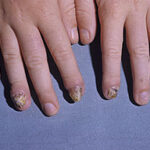
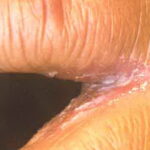
- Candidal paronychia: This is associated with frequent hand immersion in water and diabetes mellitus.
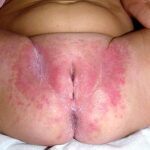
- Diaper rash: Skin irritation is exacerbated by wet diapers
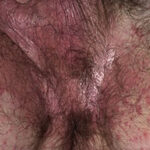
- Perianal candidiasis: Skin maceration and pruritus are frequent with frequent extension to the perineum.
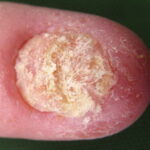
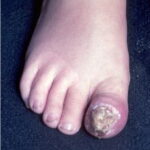
- Onychomycosis: Candida species are the most common fungal cause of this nail disease
Chronic mucocutaneous candidiasis is a term used to describe a heterogeneous group of Candida infections of the skin, mucous membranes, hair, and nails, which has a protracted course despite typical therapy. It is associated with T-cell lymphocyte dysfunction. A specific subset of patients with this phenomenon have autoimmune polyendocrinopathy-candidosis-ectodermal dystrophy (APECED) syndrome with associated endocrine disorders.
Invasive candidal infections
Invasive candidiasis is a term used to describe severe and invasive disorders that include candidemia, disseminated candidiasis, deep organ involvement, endocarditis, endophthalmitis, and meningitis. Invasive infection is also described as the isolation of Candida from a normally sterile body site, including blood, peritoneal fluid, pleural fluid, intra-articular fluid, or cerebrospinal fluid.
Risk factors for invasive candidiasis include prolonged intensive care unit (ICU) stay (incidence peaks around day 10), presence of a central venous catheter, acute renal failure, treatment with broad-spectrum antibiotics, parenteral nutrition, high APACHE II scores, diabetes, immunosuppressive therapy, surgery (especially upper gastrointestinal tract), hemodialysis, pancreatitis, malignancy, transplantation, and organ dysfunction. In one recent study Candida accounted for 45% of invasive fungal infections in renal allograft patients.13
Skin manifestations of disseminated candidiasis include clusters of painless pustules on an erythematous base on any area of the body. These lesions may be macular or pustular or may have central necrosis.
Acute disseminated candidiasis, or hepatosplenic candidiasis, is most commonly seen in patients with hematologic malignancy who recently had an episode of neutropenia. Symptoms include fever, right upper quadrant pain, and tender hepatosplenomegaly. Multiple organs are frequently involved, and discrete persistent microabscesses occur in the liver, spleen, and kidneys. A palpable erythematous rash may be present indicating evidence of small-vessel vasculitis. Presumed etiology is prior episode of candidemia, although invasion through portal vasculature has been theorized.
Central nervous system candidiasis usually occurs as a complication of hematogenously disseminated candidiasis. Candida typically forms multiple microabscesses and small macroabscesses scattered throughout the brain.14 Patients with Candida meningitis usually have meningeal irritation and CSF pleocytosis. Untreated, the mortality rate is high. Intraventricular drains increase risk of CNS candidal infection.
Candidal pneumonia occurs rarely as bronchopneumonia originating from endobronchial inoculation or more commonly a hematogenously seeded, nodular diffuse infiltrate. This presentation may be difficult to distinguish from congestive heart failure or Pneumocystis pneumonia. Diagnosis is also complicated by the inability to confirm that positive cultures are not oropharyngeal contaminant or colonization. Candida empyema cases have been documented.15
Candida can infect both the pericardium and myocardium, and these infections are usually associated with disseminated disease. Candidal pericarditis is rare but fatal without treatment and has been known to cause tamponade. Infective endocarditis with Candida is usually seen in patients with a chronic indwelling intravenous catheter or large-caliber hemodialysis catheter. Other risk factors include congenital cardiac abnormalities, prosthetic valves, and intravenous illicit drug use. Fungal vegetations are often large and more frequently associated with embolic events. The mortality rate is approximately 45% with combined medical and surgical therapy.
Urethral candidiasis can occur in both men and women. In women, it is commonly secondary to an extension of Candida vaginitis. In men, it is usually secondary to sexual contact with women with vaginitis. Invasive infections of the bladder and kidneys can occur, though it is typically in immunocompromised patients and secondary to hematogenous spread. This hematogenous spread can also lead to acute renal infarction secondary to the infiltration of the renal parenchyma and occlusion of the hilar vessels.16
Ocular candidiasis can occur in the form of endophthalmitis. Endophthalmitis may occur secondary to exogenous spread, such as trauma or surgery, or endogenous spread as a result of hematologic seeding.
Untreated candidemia has been associated with retinal lesions in up to 37% of patients. Candida ophthalmitis begins as a choroidal lesion that progresses to an area of retinal necrosis followed by vitreitis and endophthalmitis. Endophthalmitis is characterized by retinal infiltrates and vitreous abnormalities. Chorioretinal involvement appears as focal, white, infiltrative lesions on the retina. Vitreal haze is present with vitreous extension of the infection. Symptoms include pain and decreased visual acuity. Untreated, ophthalmitis will lead to blindness. Typically, involvement is unilateral, but bilateral cases have been reported. C albicans is the most frequent culprit.
Osseous or intra-articular infections may occur secondary to either hematogenous spread or exogenous inoculation during trauma or joint injection. Osteomyelitis occurs most commonly in vertebrae in adults and in long bones in children.17,18 Spinal infection can progress to a diskitis. Arthritis can be acute and suppurative, and the knee is most commonly affected.19 Diagnosis of osteoarticular infections may be delayed as symptoms are frequently more subtle than bacterial infections in the same location and patients often present several weeks to months after an episode of candidemia. Fever is typically absent.
Candidal peritonitis is most frequently secondary to peritoneal dialysis catheter seeding or gastrointestinal surgery.
Mediastinitis secondary to candidal infection may occur after thoracic surgery.
Neonatal invasive candidiasis occurs with an incidence inversely proportional to birth weight. Candida colonization is found in approximately 30% of infants weighing less than 1500 grams at birth weight. Sources of invasive infection in one study included blood (70%), urine (15%), cerebrospinal fluid (10%), and peritoneal fluid (5%). C albicans and C parapsilosis are the most common species found in neonates.
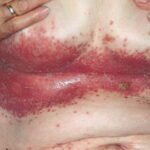
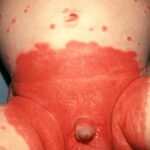
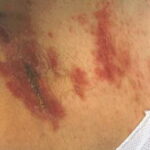
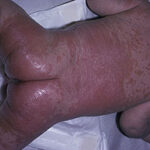
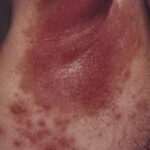
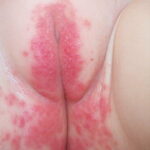
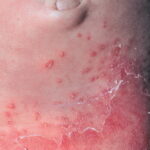
Candida amnionitis may occur after prolonged rupture of the membranes in mothers given parenteral antibiotics. A neonate’s skin may have pustules, vesicles, or diffuse erythema (shown in the image below). Neonates can also develop candidemia even after cesarean delivery due to premature rupture of amniotic membranes.20Causes
- C albicans is the most common pathogenic species identified. Other species that are commonly found include C glabrata, C parapsilosis, C tropicalis, and C krusei.
- Non-albicans species have been associated with specific patient populations. C glabrata is seen most commonly in patients with neoplastic disease; C tropicalis in patients with leukemia or neutropenia; C parapsilosis among neonates and those with an indwelling intravenous catheter; and C krusei in stem cell transplant recipients and in patients with leukemia who have received fluconazole prophylaxis.
- Denture use, immunosuppressant, antibiotic therapy, and aging are risk factors for oral colonization with C glabrata. C glabrata exhibits lower oral keratinocyte-adherence capacity but higher denture surface adherence ability.
Laboratory Studies
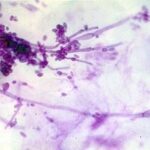
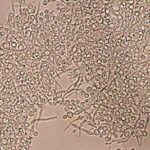
- The criterion standard diagnostic tool for mucocutaneous candidiasis is culture. Cutaneous or mucosal scrapings can be used for a potassium hydroxide smear or Gram stain, which show hyphae, pseudohyphae, and budding yeast forms. The sensitivity of wet mount is as low as 39.6%.
- Savyon Diagnostics currently is developing a rapid yeast detection kit for home diagnosis of vulvovaginal candidiasis. Preliminary data indicate better sensitivity than wet mount, and it costs less than culture.
- Invasive Candida infections are typically difficult to diagnose because the clinical presentation is frequently similar to other disease states or bacterial infections (eg, blood, urine, CSF), and cultures are unreliable. In one report, blood cultures were positive in only 50-70% of disseminated candidiasis cases when the disease was proven by autopsy. Thus, nonculture diagnostic techniques are frequently used to aid in diagnosis. The 1,3 beta-glucan assay, which measures the fungal cell was component, has a sensitivity of 70% and specificity of 87%.
- Non–Food Drug Administration (FDA)–approved diagnostic tools include the Platellia Aspergillus ELISA (PA-ELISA) and polymerase chain reaction–based assay.
- The peptide nucleic acid fluorescent in situ hybridization (PNA-FISH) is used to distinguish C albicans from non-albicans Candida species.
- CHROMagear is a specialized media for Candida isolation, which distinguishes C albicans, C tropicalis, and C krusei based on the species’ distinctive pigments.
Imaging Studies
- Imaging studies are not necessary for routine mucocutaneous candidiasis.
- Radiographic evaluations for systemic candidiasis are often nonspecific; thus, differentiation of candidiasis from other disease processes may be difficult.
- Chest radiography may be helpful in making the diagnosis of pulmonary candidiasis.
- Disease spreads via the airway system. Chest radiographic findings are characterized by bilateral, diffuse, and poorly marginated areas; pulmonary parenchymal densities are common.
- Other nonspecific findings include air bronchogram and obscure cardiac and hemidiaphragm borders.
- Hematogenous infection may produce a miliary nodular pattern.
- Esophagography with contrast is indicated for diagnosis of esophagitis caused by Candida species.
- Peristaltic abnormalities caused by small plaques appear as superficial filling defects. A nodular or cobblestone pattern may be seen.
- Findings may be similar to those seen with esophagitis caused by cytomegalovirus (CMV) or herpes simplex virus (HSV).
- Stricture may occur in severe esophageal candidiasis.
- Ultrasonography is useful for diagnosis of microabscesses in the liver, spleen, or kidneys.
- “Wheel-within-a-wheel” hypoechoic zones surrounded by hyperechoic zones are early findings.
- Typical “bull’s-eye” lesion may evolve from the initial lesion.
- A uniformly hyperechoic lesion may be observed.
- Echogenic foci with variable degrees of acoustic shadowing are late findings.
- CT scan allows diagnosis of microabscesses, represented by low-attenuation foci, in the liver, spleen, or kidneys.
- Intravenous pyelography (IVP) may be helpful in the diagnosis of urinary tract candidiasis.
- Renal edema may be present from multiple microabscesses, deformity of renal outline, diminished renal excretion, papillary necrosis, and candidal fungal balls in the bladder or collecting system may be seen.
- Hydronephrosis is a common late finding.
- CT scan/MRI is indicated for evaluation of CNS lesions and prior to lumbar puncture.
Procedures
Endoscopy, tissue biopsy, and percutaneous needle aspiration of a body site suspected of Candida infection are recommended to aid in diagnosis. Positive candidal cultures in a normally sterile site should not be disregarded as a contaminant.
Emergency Department Care
Mucocutaneous candidiasis is often encountered and treatment initiated in the emergency department. Systemic infections in patients with risk factors for Candida infection should be admitted to the hospital and cultures taken prior to initiating antimicrobial therapy
Other treatment of candidiasis- Generally, the echinocandin class of antifungals should be used as the first-line treatment of candidemia (ie, the critically ill patients or patients with prior azole treatment). There is already a report of echinocandin resistance after long-term therapy for candidemia.21
- Polyenes should not be used to treat patients who have renal failure, and echinocandins and azoles should not be used in patients with severe liver disease because of their respective side effect profiles and pharmacokinetic properties.22
- Mucocutaneous infection typically responds to topical therapy. Nystatin is the least expensive option for oral thrush, but patients frequently complain of its bitter taste. Clotrimazole troches are as effective and less bitter. Proper denture cleansing and care are important measures against oral candidiasis.
- Topical therapy is first line for esophageal candidiasis, but systemic therapy is typically required.
- Intertrigo and diaper rash respond to decreased moisture around the skin. Nystatin powder or cream is used with the addition of a topical steroid for diaper rash.
- Uncomplicated vulvovaginal candidiasis treatment includes many options of topical or oral therapy. Recurrent candidal vaginitis requires a prolonged course of oral medication; probiotic Lactobacillus may help in facilitating treatment of this disease.23
- Invasive candidiasis typically requires parenterally administered antifungal therapy.
- Candida endocarditis frequently requires both medical and surgical therapy. Valve replacement and vegetation removal is often necessary. Antifungal therapy is typically continued for 6-10 weeks parenterally.
- CNS candidal infection can often successfully be treated without intrathecal instillation.
- Peritoneal candidal infection secondary to peritoneal dialysis may respond to peritoneal infusion of antifungal agents in dialysate fluid.
- Candida keratitis may require corneal grafting if not responsive to treatment.
- Full-term infants with Candida amnionitis typically respond to topical therapy. Premature infants frequently require systemic antifungal agents and have a poorer prognosis.
- Endophthalmitis may require vitrectomy and direct intravitreal antifungal instillation.
Antifungal therapy should be started immediately after necessary cultures have been obtained from all suspected sites of infection.
Significant advances have been made in the treatment of Candida with the development of newer azoles, echinocandins, and lipid formulations of amphotericin B.
Antifungals, azole
Azoles are fungicidal only at very high concentrations. Azoles function by selectively inhibiting the synthesis of an essential component of fungal cell membrane, ergosterol.
Fluconazole (Diflucan)
Synthetic oral antifungal (broad-spectrum bistriazole) that selectively inhibits fungal cytochrome P-450 and sterol C-14 alpha-demethylation, which prevents conversion of lanosterol to ergosterol, thereby disrupting cellular membranes. Has little affinity for mammalian cytochromes, which is believed to explain its low toxicity. Available as tablets for oral administration, as a powder for oral suspension, and as a sterile solution for IV use. Has fewer adverse effects and better tissue distribution than older systemic imidazoles.
Daily dose varies with indication.
Fluconazole penetrates the cerebrospinal fluid, kidneys, and liver well. It is concentrated and excreted by the kidneys in its active form so is effective against urinary tract infections.Adult
Oropharyngeal and esophageal disease: 100 mg/d PO/IV for 7-14 d
Candidemia and invasive candidiasis: 800-mg loading dose, followed by 400 mg/d PO/IV; if C glabrata, dose should be 800 mg/d
Renal insufficiency:
CrCl 25-49 mL/min: Decrease dose by 50%
CrCl <25 mL/min: Decrease dose by additional 75%
Hemodialysis: Usual daily dose after each dialysis
Bone marrow transplant: 200-400 mg/d PO/IV starting at time of bone marrow ablation and continuing until neutropenia resolves; recent studies seem to indicate continuing therapy for longer periods of time may decrease mortality in transplant recipientsPediatric
Candidemia: 10-12 mg/kg/d PO/IV loading dose followed by 4.5-6 mg/kg/d PO/IV
CYP450 2C19 and 3A4 inhibitor; levels may increase with hydrochlorothiazide; fluconazole levels may decrease with long-term coadministration of rifampin; may increase concentrations of theophylline, phenytoin, tolbutamide, cyclosporine, glyburide, and glipizide; effects of anticoagulants may increase with fluconazole coadministration
Documented hypersensitivity
Pregnancy
C – Fetal risk revealed in studies in animals but not established or not studied in humans; may use if benefits outweigh risk to fetus
Precautions
Adjust dose for renal insufficiency; closely monitor if rashes develop, and discontinue drug if lesions progress; may cause clinical hepatitis, cholestasis, and fulminant hepatic failure (including death) when taken with underlying medical conditions (eg, AIDS, malignancy) or while taking multiple concomitant medications; not recommended for breastfeeding mothers; QT prolongation and torsade de pointes can occur; rarely, anaphylactic reactions, Stevens-Johnson syndrome, and toxic necrolysis can occur
Convenience and efficacy of single-dose regimen for treatment of vaginal yeast infections should be weighed against difficulties resulting from higher incidence of adverse reactions reported with oral fluconazole versus intravaginal agentsTerconazole vaginal (Terazol-3, Terazol-7)
Damages fungal cell wall membrane by inhibiting biosynthesis of ergosterol. Membrane permeability is increased, causing nutrients to leak out, resulting in fungal cell death.
Adult
Vaginal candidiasis:
80-mg tab: Insert 1 suppository (2.5 g) intravaginally hs for 3 d
0.4% cream: Insert 1 applicatorful (5 g) intravaginally hs for 7Pediatric
Not established
None reported
Documented hypersensitivity
Pregnancy
C – Fetal risk revealed in studies in animals but not established or not studied in humans; may use if benefits outweigh risk to fetus
Precautions
If sensitivity or chemical irritation occurs, discontinue use; use only externally; avoid contact with eyes
Butoconazole (Femstat-3, Gynazole-1)
Broad-spectrum antifungal agent that inhibit yeast growth by altering cell membrane permeability, which causes fungal cell death.
Use 2% vaginal cream. Available over the counter.Adult
Vaginal candidiasis: Insert 1 applicatorful (5 g) intravaginally hs for 3 d
Pediatric
Not established
None reported
Documented hypersensitivity
Pregnancy
C – Fetal risk revealed in studies in animals but not established or not studied in humans; may use if benefits outweigh risk to fetus
Precautions
If sensitivity or chemical irritation occurs, discontinue use; use only externally; avoid contact with eyes
Voriconazole (VFEND)
Available in oral and parenteral forms. FDA approved for esophageal candidiasis and candidemia. Used clinically for serious candidal infections refractory to amphotericin B. Voriconazole has been found to be active against C glabrata and C krusei as well as isolates that have developed resistance to fluconazole.
Adult
Loading dose: 6 mg/kg IV q12h infused over 2 h for 2 doses
Maintenance: 4 mg/kg IV q12h infused over 2 h, when able to tolerate PO may switch to 200 mg PO q12h
Note: For inadequate response, may increase to 300 mg PO q12h; <40 kg administer oral maintenance dose of 100 mg PO q12h (may increase to 150 mg PO q12h)Pediatric
Not established
CYP450 2C19 (highest affinity), 2C9, and 3A4 (minor) substrate and inhibitor; CYP450 inducers (eg, rifampin) have shown to decrease steady-state peak plasma levels by up to 93%; may increase serum levels of drugs metabolized by CYP450 2C19 or 2C9, of which some are contraindicated (eg, sirolimus, pimozide, quinidine, cisapride, ergot alkaloids), others may need more frequent monitoring (eg, cyclosporine, tacrolimus, warfarin, HMG-CoA inhibitors, benzodiazepines, calcium channel blockers)
Documented hypersensitivity; do not administer IV form with CrCl <50 mL/min (decreased excretion of IV vehicle); coadministration with rifampin, rifabutin, carbamazepine, barbiturates, sirolimus, pimozide, quinidine, cisapride, ergot alkaloids
Pregnancy
D – Fetal risk shown in humans; use only if benefits outweigh risk to fetus
Precautions
Decrease maintenance dose with hepatic dysfunction; common adverse effects include visual disturbances, fever, rash, vomiting, nausea, diarrhea, headache, sepsis, peripheral edema, abdominal pain, rash (including Stevens-Johnson syndrome and phototoxicity), and respiratory disorder; rare cases of severe hepatotoxicity have been reported; administer PO 1 h ac or pc
Ketoconazole (Nizoral)
Imidazole broad-spectrum antifungal agent. Nizoral impairs synthesis of ergosterol (the main sterol of fungal cell membranes), allowing increased permeability and leakage of cellular components, causing cell death. Used in treatment of chronic mucocutaneous candidiasis and cutaneous infections.
Adult
Mucocutaneous infections: 400 mg PO qd (with food) for 3-9 mo
Cutaneous infections: Apply topically qd for 14 dPediatric
Mucocutaneous infections:
<2 years: Not recommended
>2 years: 5 mg/kg/d PO once; not to exceed 800 mg qdIsoniazid may decrease bioavailability; coadministration of rifampin may decrease effects of either; may increase effect of anticoagulants; may increase toxicity of corticosteroids and cyclosporine (cyclosporine dosage can be adjusted); may decrease theophylline levels
Documented hypersensitivity; fungal meningitis
Pregnancy
C – Fetal risk revealed in studies in animals but not established or not studied in humans; may use if benefits outweigh risk to fetus
Precautions
Hepatotoxicity may occur; may reversibly decrease corticosteroid serum levels (adverse effects avoided with dose of 200-400 mg/d); administer antacid, anticholinergics, or H2-blockers at least 2 h after taking ketoconazole
Itraconazole (Sporanox)
Has fungistatic activity. Synthetic triazole antifungal agent that slows fungal cell growth by inhibiting cytochrome P-450–dependent synthesis of ergosterol, a vital component of fungal cell membranes. Effective against broad range of fungi, including Candida species and is indicated for the treatment of cutaneous, oral, esophageal, and disseminated candidiasis.
Available in IV, 100-mg capsules, and oral solution at 10 mg/mL.
Capsules require gastric acidity for absorption and should be taken with food to increase absorption. Liquid formulation increases bioavailability and decreases need for acidity for proper absorption.
Use of solution has been recommended in mucosal and invasive candidiasis, while capsules can be used in onychomycosis and dermatophyte infections.Adult
Cutaneous candidiasis and onychomycosis: 200 mg PO bid for 7 d/mo for 3-6 mo
Oropharyngeal and esophageal candidiasis: 200 mg/d PO for 7-14 d
Candidemia and invasive candidiasis: 200 mg PO tid for 3 d, followed by 200 mg bid for 14-21 dPediatric
Cutaneous candidiasis: 3-5 mg/kg/d PO for 30 d
Antacids may reduce absorption of itraconazole; edema may occur with coadministration of calcium channel blockers (eg, amlodipine, nifedipine); hypoglycemia may occur with sulfonylureas; rhabdomyolysis may occur with coadministration of HMG-CoA reductase inhibitors (lovastatin or simvastatin); coadministration with cisapride can cause cardiac rhythm abnormalities and death
Monitoring of liver enzymes recommended; potent inhibitor of CYP3A4 (may increase plasma concentrations of terfenadine, midazolam, cyclosporin, cisapride, quinidine, atorvastatin, simvastatin, lovastatin when coadministered); CYP3A4 inducers, such as phenytoin, rifampin, and isoniazid, may decrease itraconazole concentrationsDocumented hypersensitivity
Pregnancy
C – Fetal risk revealed in studies in animals but not established or not studied in humans; may use if benefits outweigh risk to fetus
Precautions
Caution in hepatic insufficiency