Acute sunburn= حرق شمسي حاد
Acute sunburn
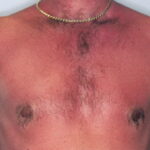
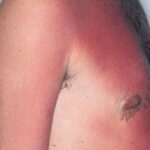
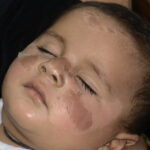
Sunburn is an acute cutaneous inflammatory reaction that follows excessive exposure of the skin to ultraviolet radiation (UVR). UVR exposure can come from a variety of sources, including sun, tanning beds, phototherapy lamps, and arc lamps.1 Long-term adverse health effects of repeated exposure to UVR are well described but are beyond the scope of this article. Sunburn is generally classified as a superficial or first-degree burn
Pathophysiology
Exposure to solar radiation has the beneficial effects of stimulating the cutaneous synthesis of vitamin D and providing radiant warmth. Unfortunately, when the skin is subjected to excessive radiation in the ultraviolet range, deleterious effects may occur. The most conspicuous is acute sunburn or solar erythema.2
Initially, UVR causes vasodilation of cutaneous blood vessels, resulting in the characteristic erythema. Within an hour of UVR exposure, mast cells release preformed mediators including histamine, serotonin, and tumor necrosis factor, leading to prostaglandin and leukotriene synthesis.2,3 Cytokine release additionally contributes to the inflammatory reaction, leading to an infiltrate of neutrophils and T lymphocytes.4 Within 2 hours after UV exposure, damage to epidermal skin cells is seen. Both epidermal keratinocytes (“sunburn cells”) and Langerhans cells undergo apoptotic changes as a consequence of UVR-induced DNA damage.5,6 Erythema usually occurs 3-
Less intense or shorter-duration exposure to UVR results in an increase in skin pigmentation, known as tanning, which provides some protection against further UVR-induced damage.8 The increased skin pigmentation occurs in 2 phases: (1) immediate pigment darkening and (2) delayed tanning. Immediate pigment darkening occurs during exposure to UVR and results from alteration of existing melanin (oxidation, redistribution). It may fade rapidly or persist for several days. Delayed tanning results from increased synthesis of epidermal melanin and requires a longer period of time to become visible (24-72 h). With repeated exposure to UVR, the skin thickens, primarily due to epidermal hyperplasia with thickening of the stratum corneum. UVR exposure also suppresses cutaneous cell-mediated immunity, contributing to nonmelanoma skin cancer and infectious disease development
4 hours after exposure, with peak levels at 24 hours
History
History and symptoms for sunburn may include the following:
- Recent sun exposure or outdoor activity; outdoor occupations or hobbies; use of indoor tanning equipment
- Erythema develops after 3-4 hours and peaks at 12-24 hours.7
- Pain
- Possible fever, chills, malaise, nausea, or vomiting in severe cases
- Blistering
- Erythema that resolves over 4-7 days, usually with skin scaling and peeling7
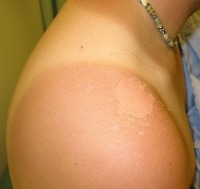
Subacute sunburn of shoulder with peeling in a 21-year-old male.
[ CLOSE WINDOW ]Subacute sunburn of shoulder with peeling in a 21-year-old male.
- Assess for exposure to photosensitizing drugs. See eMedicine article Drug-Induced Photosensitivity for an in-depth discussion and list of common photosensitizing drugs.
- Assess for heavy alcohol use, which is associated with sunburning.10,15
Physical
- Patients at highest risk typically have fair skin, blue eyes, and red or blond hair.13
- The acute inflammatory response is greatest 12-24 hours after exposure.7
- Erythema
- Warmth
- Tenderness
- Edema
- Blistering (severe cases), a sign of either a superficial partial-thickness or deep partial-thickness (second-degree) burn16
- Fever can present in severe cases.7
- UVR may be transmitted through clothing, especially when wet, so sunburn may occur under clothed skin.17
- Delayed scaling and desquamation occurs 4-7 days after exposure.7
Causes
Sunburn is caused by excessive exposure of the skin to UVR. The ultraviolet spectrum can be divided into ultraviolet A-I (UVA-I), 340-400 nm; ultraviolet A-II (UVA-II), 320-340 nm, ultraviolet B (UVB), 290-320 nm; and ultraviolet C (UVC), 200-290 nm.1 Solar UVR of wavelengths shorter than 290 nm are filtered out or absorbed in the outer atmosphere and are not encountered at sea level.1 Shorter wavelength UVB rays are much more effective at inducing erythema than UVA rays and, therefore, are the principal cause of sunburn.1 However, UVA comprises the majority of UVR reaching the surface of the earth (about 95-98% at midday) and, therefore, accounts for a significant percentage of the immediate and long-term cutaneous effects of UVR.1
The minimal single dose of UVR (energy per unit area) required to produce erythema after 24 hours at an exposed site is known as the minimal erythema dose (MED). This dose differs by skin type.2
Multiple factors influence UVR-induced erythema; these are listed below.
- Wavelength: UVB is more erythemogenic than UVA.2
- Skin type/pigmentation: Compared with type I-II skin, patients with type IV-V skin require 3-5 times more UVR exposure to cause erythema.13
- Hydration: UVR causes erythema in moist skin more effectively than dry skin.18
- Environmental reflection: Radiation is 80% reflected by snow and ice, compared with 15% by sand.12
- Ozone coverage: Increased levels of ozone filter out more UVR.12
- Altitude: Thinner atmosphere at higher altitudes absorbs less UVR.12
- Latitude: Exposure is greater nearer the equator.12
- Time of day: UVR exposure is greatest from 10 am to 4 pm, when the sun is highest in the sky
Prehospital Care
- In most cases, prehospital care involves providing simple first aid to treat patient symptoms.
- In severe cases, patients may develop second-degree burns, which could require aggressive fluid resuscitation and skin care.
Emergency Department Care
- Most sunburns, while painful, are not life threatening, and treatment is primarily symptomatic.7
- Nonsteroidal anti-inflammatory drugs (NSAIDs) have antiprostaglandin effects and are useful to relieve pain and inflammation, especially when given early. Cool soaks with water or Burrow solution (aluminum acetate solution) also provide temporary relief.7,19
- Fluid replacement (oral or intravenous) for severe erythema or concomitant fluid loss.20
- Studies of emollients such as aloe vera have failed to demonstrate decreased recovery times, but these treatments may help with sunburn symptoms.21
- Systemic steroids are sometimes used to shorten the course and to reduce the pain of sunburn when given early and in relatively high doses (equivalent to 40-60 mg/d of prednisone).7 Although this is described in the literature, currently, there is no evidence to support this practice.21
- Prescribe steroids for only a few days, with no need for a taper.
- In the presence of partial-thickness (second-degree) burn, steroids are best avoided because they increase the risk of infection.
- Topical steroids show minimal, if any, benefit
Medication
The symptoms of minor sunburn can be relieved to some extent with cool compresses or a cool bath. Administration of nonprescription analgesics and NSAIDs for the treatment of pain and inflammation is recommended.
Analgesic Nonsteroidal Anti-inflammatory Drug
These medications can reduce the pain and inflammation associated with sunburn.
Naproxen (Aleve, Anaprox, Naprelan, Naprosyn)
For relief of mild to moderate pain; inhibits inflammatory reactions and pain by decreasing activity of cyclooxygenase, which results in a decrease of prostaglandin synthesis.
- Dosing
- Interactions
- Contraindications
- Precautions
Adult
500 mg PO followed by 250 mg q6-8h; not to exceed 1.25 g/d
Pediatric
<2 years: Not established
>2 years: 2.5 mg/kg/dose PO; not to exceed 10 mg/kg/d- Dosing
- Interactions
- Contraindications
- Precautions
Coadministration with aspirin increases risk of inducing serious NSAID-related adverse effects; probenecid may increase concentrations and, possibly, toxicity of NSAIDs; may decrease effect of hydralazine, captopril, and beta-blockers; may decrease diuretic effects of furosemide and thiazides; may increase PT when taking anticoagulants (instruct patients to watch for signs of bleeding); may increase risk of methotrexate toxicity; phenytoin levels may be increased when administered concurrently
- Dosing
- Interactions
- Contraindications
- Precautions
Documented hypersensitivity; peptic ulcer disease; recent GI bleeding or perforation; renal insufficiency
- Dosing
- Interactions
- Contraindications
- Precautions
Pregnancy
B – Fetal risk not confirmed in studies in humans but has been shown in some studies in animals
D – Fetal risk shown in humans; use only if benefits outweigh risk to fetus
Precautions
Acute renal insufficiency, interstitial nephritis, hyperkalemia, hyponatremia, and renal papillary necrosis may occur; patients with preexisting renal disease or compromised renal perfusion risk acute renal failure; leukopenia occurs rarely, is transient, and usually returns to normal during therapy; persistent leukopenia, granulocytopenia, or thrombocytopenia warrants further evaluation and may require discontinuation of drug
Aspirin (Bayer, Anacin, Bufferin)
Used for the treatment of mild to moderate pain. Also acts on the hypothalamus heat-regulating center to reduce fever.
- Dosing
- Interactions
- Contraindications
- Precautions
Adult
650 mg PO bid/qid; not to exceed 4 g/d in equally divided doses
Pediatric
10-15 mg/kg/dose PO q4-6h; not to exceed 60-80 mg/kg/d
- Dosing
- Interactions
- Contraindications
- Precautions
Effects may decrease with antacids and urinary alkalinizers; corticosteroids decrease salicylate serum levels; additive hypoprothrombinemic effects and increased bleeding time may occur with coadministration of anticoagulants; may antagonize uricosuric effects of probenecid and increase toxicity of phenytoin and valproic acid; doses >2 g/d may potentiate glucose-lowering effect of sulfonylurea drugs
- Dosing
- Interactions
- Contraindications
- Precautions
Documented hypersensitivity; liver damage; hypoprothrombinemia; vitamin K deficiency; bleeding disorders; asthma; children (<16 y) with flu (because of association with Reye syndrome)
- Dosing
- Interactions
- Contraindications
- Precautions
Pregnancy
C – Fetal risk revealed in studies in animals but not established or not studied in humans; may use if benefits outweigh risk to fetus
D – Fetal risk shown in humans; use only if benefits outweigh risk to fetus
Precautions
May cause transient decrease in renal function and aggravate chronic kidney disease; avoid use in patients with severe anemia, in those with history of blood coagulation defects, or in those taking anticoagulants
Ibuprofen (Advil, Motrin, Nuprin)
Usually the DOC for the treatment of mild to moderate pain, if no contraindications are present.
- Dosing
- Interactions
- Contraindications
- Precautions
Adult
200-400 mg PO q4-6h while symptoms persist; not to exceed 3.2 g/d
Pediatric
30-70 mg/kg/d PO tid/qid
- Dosing
- Interactions
- Contraindications
- Precautions
Coadministration with aspirin increases risk of inducing serious NSAID-related adverse effects; probenecid may increase concentrations and, possibly, toxicity of NSAIDs; may decrease effect of hydralazine, captopril, and beta-blockers; may decrease diuretic effects of furosemide and thiazides; monitor PT closely (instruct patients to watch for signs of bleeding); may increase risk of methotrexate toxicity; phenytoin levels may be increased when administered concurrently
- Dosing
- Interactions
- Contraindications
- Precautions
Documented hypersensitivity; peptic ulcer disease; recent GI bleeding or perforation; renal insufficiency; high risk of bleeding
- Dosing
- Interactions
- Contraindications
- Precautions
Pregnancy
B – Fetal risk not confirmed in studies in humans but has been shown in some studies in animals
D – Fetal risk shown in humans; use only if benefits outweigh risk to fetus
Precautions
Caution in congestive heart failure, hypertension, and decreased renal and hepatic function; caution in anticoagulation abnormalities or during anticoagulant therapy
Analgesic, Miscellaneous
These agents are used to decrease the pain associated with sunburn.
Acetaminophen (Tylenol, Aspirin-Free Anacin, Feverall, Tempra)
DOC for pain in patients with documented hypersensitivity to aspirin or NSAIDs, in those with upper GI disease, or in those who are taking oral anticoagulants.
Effective in relieving mild to moderate acute pain; however, has no peripheral anti-inflammatory effects. May be preferred in elderly patients because of fewer GI and renal side effects.- Dosing
- Interactions
- Contraindications
- Precautions
Adult
325-650 mg PO/PR q4-6h or 1000 mg tid/qid; not to exceed 4 g/d
Pediatric
<12 years: 10-15 mg/kg/dose PO q4-6h prn; not to exceed 2.6 g/d
>12 years: 325-650 mg PO q4h; not to exceed 4 g/d- Dosing
- Interactions
- Contraindications
- Precautions
Rifampin can reduce analgesic effects of acetaminophen; coadministration with barbiturates, carbamazepine, hydantoins, and isoniazid may increase hepatotoxicity
- Dosing
- Interactions
- Contraindications
- Precautions
Documented hypersensitivity; known G-6-PD deficiency
- Dosing
- Interactions
- Contraindications
- Precautions
Pregnancy
B – Fetal risk not confirmed in studies in humans but has been shown in some studies in animals
Precautions
Hepatotoxicity possible in chronic alcoholics following various dose levels; severe or recurrent pain or high or continued fever may indicate a serious illness; contained in many OTC products and combined use with these products may result in toxicity due to cumulative doses exceeding recommended maximum dose
Corticosteroids
Because they modify the body’s immune response, corticosteroids are thought to decrease erythema and shorten the course of sunburn.
Prednisone (Deltasone, Orasone, Meticorten)
May decrease inflammation by reversing increased capillary permeability and suppressing PMN activity.
- Dosing
- Interactions
- Contraindications
- Precautions
Adult
40-60 mg PO qd
Pediatric
1 mg/kg PO qd
Deterrence/Prevention
- Prevention is the most effective therapy for sunburn. Individual and community educational programs can be effective in decreasing overall sun exposure or increasing use of sunscreen or protective clothing.23,24
- Avoid sun exposure, especially during the period of peak solar radiation (from 10 am to 4 pm).9
- Regularly use sunscreen with an adequate SPF for a given skin type.
- SPF is the ratio of the amount of UV energy needed to produce erythema on protected skin to the amount of UV energy needed to produce erythema on unprotected skin.25
- Refer to the Table for recommended sunscreen levels for everyday protection and outdoor activity protection.
- Apply at least 30 minutes prior to sun exposure, and reapply every 2-3 hours or after swimming, sweating, or toweling off.25
- Apply for young children prior to exposure.
- Use waterproof sunscreen when swimming or perspiring heavily.25
- Apply at least 2 mg/cm2 of sunscreen to achieve the advertised SPF (about 30 mL is adequate coverage for an average adult’s entire body). Most people apply one fifth of this amount.26
- Physical barriers (eg, zinc oxide, titanium dioxide) provide excellent protection against UVA and UVB and are photostable.14
- Chemical barriers are used in most sunscreens. Para-aminobenzoic acid (PABA) and PABA esters, UVB blockers, have fallen out of favor because of high rates of associated contact dermatitis and clothing staining. Other chemical UVB blocking agents include cinnamates and salicylates.14
- Chemical UVA blockers include avobenzone (Parsol 1789) and the recently FDA-approved drometrizole trisiloxane and terephthalylidene (Mexoryl).27
- Wear protective clothing, including wide-brimmed hat or sun visor. Clothing can be treated with over-the-counter products to increase the SPF.17
- Specialized sun-protective clothing is available and usually states the SPF each garment affords