Amyloidosis-macular=الداء النشواني البقعي
macular Amyloidosis
Amyloidosis is a generic term that signifies the abnormal extracellular tissue deposition of one of a family of biochemically unrelated proteins that share certain characteristic staining properties, including apple-green birefringence of Congo red–stained preparations viewed under polarizing light. Under electron microscopy (EM), amyloid deposits are composed of linear, nonbranching, aggregated fibrils that are 7.5-10 nm thick of indefinite length arranged in a loose meshwork.1
X-ray diffraction crystallography and infrared spectroscopy reveal that these fibrils have a meridional, antiparallel, beta-pleated sheet configuration, with polypeptide chains arranged perpendicular to the long axis of the fibrils.
Amyloid deposits contain (in addition to the fibrillar component) a nonfibrillar protein referred to as amyloid-P (Am-P). This protein is identical to normal plasma globulin, known as serum amyloid-P (SAP). Am-P constitutes 14% of the dry weight of amyloid. This protein is also found in the microfibrillar sheath of elastic fibers. SAP is closely related to the acute phase reactant C-reactive protein (CRP) and has been shown to be an elastase inhibitor.2
The SAP and the beta-pleated sheet configurations are thought to protect amyloid deposits from degradation and phagocytosis, leading to persistence of the deposits.
Macular amyloidosis has been reported in association with Sipple syndrome. The cardinal triad of this autosomal dominant syndrome is medullary thyroid carcinoma, pheochromocytoma, and hyperparathyroidism.
Pathophysiology
Amyloid deposits in macular amyloidosis and lichen amyloidosis bind to antikeratin antibodies. These deposits contain sulfhydryl groups pointing to altered keratin as a source for these deposits. Apaydin et al found no differences in staining characteristics of cytokeratins between macular amyloidosis and lichen amyloidosis.3 Interestingly, in their study, all the cytokeratins detected in amyloid deposits were of basic type (type II). This may be because, in amyloidogenesis, acidic cytokeratins such as cytokeratin 14 are degraded faster than basic types.
The exact origin of amyloid deposits in macular amyloidosis has not been determined. Two theories have been proposed to explain the origin of the amyloid deposits. These theories are not mutually exclusive, and both could be possible.
Fibrillar body theory
This theory proposed by Hashimoto suggests that the necrotic epidermal cells (colloid bodies) are transformed into amyloid by dermal macrophages and fibroblasts by a process called filamentous degeneration. The absence of amyloid deposits in other dermatoses with colloid bodies (eg, lichen planus) is explained by the brisk inflammatory reaction clearing them promptly in lichen planus, while the lack of inflammatory cells leads to the formation of amyloid deposits in macular amyloidosis.4,5 This theory does not explain how the alpha type of keratin tertiary structure is degraded and converted into the beta-pleated sheet configuration of amyloid.
History
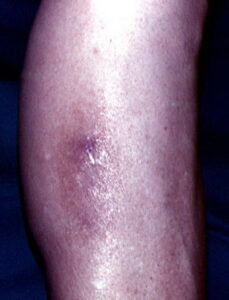
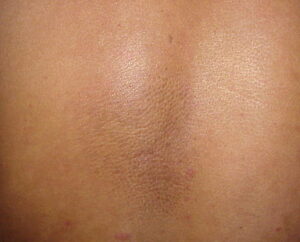
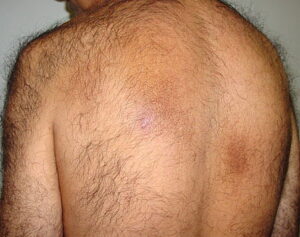
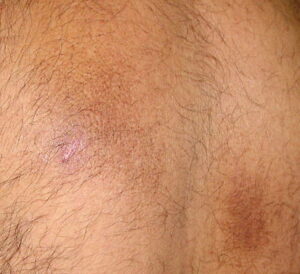
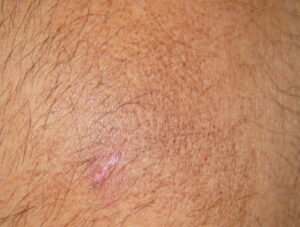
Macular amyloidosis is a pruritic eruption that is variable in severity. Frequently, patients seek medical attention because of the hyperpigmentation.
Physical
Macular amyloidosis is a pruritic eruption consisting of small, dusky-brown or grayish pigmented macules distributed symmetrically over the upper back and, in some patients, the arms.
Although a reticulated or rippled pattern of pigmentation has been emphasized as a characteristic and diagnostic feature of macular amyloidosis, in 2 case series, less than 50% of patients had this feature.
Causes
Constant friction and rubbing with a nylon brush or towel may cause macular amyloidosis
Histologic Findings
Many stains can demonstrate amyloid deposits in the skin. The best known is the Congo red stain, which under polarizing light gives apple-green birefringence. Other stains include periodic acid-Schiff (PAS); methyl violet; crystal violet; various cotton dyes (eg, pagoda red, Sirius red); and the fluorescent dyes, thioflavin-T and Phorwhite BBU.
In macular amyloidosis, the amyloid deposits are usually found within the dermal papillae. The amyloid deposits are usually globular, resembling colloid bodies, and they may be in contact with basal cells at the dermoepidermal junction. The deposits can be minute, escaping detection. For this reason, macular amyloidosis is part of the differential diagnosis for the “normal skin” slide, sometimes called invisible dermatosis. Minimal epidermal changes, such as hyperkeratosis and hypergranulosis, are occasionally observed
Medical Care
- Because of the growing appreciation of the importance of pruritus as the primary trigger for the deposition of amyloid, treatment modalities are directed toward the relief of pruritus in macular amyloidosis and lichen amyloidosis.
- Sedating antihistamines have been found to be moderately effective.
- Topical dimethyl sulfoxide (DMSO), a chemical solvent, and intralesional steroids are beneficial if combined with other modalities. DMSO has been used with moderate success, but failures have also been reported.9,10,11 Pandhi et al and Lim et al reported a lack of effect with DMSO treatment for cutaneous amyloidosis.12,13
- Treatment with ultraviolet B (UV-B) light can provide symptomatic relief.
- Sawamura et al reported satisfying improvement of lichen amyloidosis with pulsed dye laser therapy. Both pruritus and the papular eruption of lichen amyloidosis improved.14
Surgical Care
- Aggressive strategies proposed for the removal of amyloid include laser vaporization, dermabrasion, and excision of individual lesions. However, lesions and pruritus usually promptly recur after these treatments.
- Electrodesiccation and curettage provided an acceptable result in one report.15
- In a prospective, side-by-side, controlled, clinical trial study, Ostovari et al used the Q-switched Nd:YAG laser (532 nm and 1064 nm) in 20 subjects with a clinical diagnosis and pathology confirmation of macular amyloidosis. Using colorimetric score assessment and digital photographs before laser therapy and 8 weeks after treatment, they concluded that the 2 lasers are effective in reducing the degree of macular amyloidosis pigmentation, with the 532-nm laser being more effective than the 1064-nm laser. The pictures shown in this paper were of low quality.8
Medication
The goal of pharmacotherapy is to reduce morbidity.
Antihistamines
These agents act by competitive inhibition of histamine at the H1 receptor. They may control itching by blocking effects of endogenously released histamine.
Chlorpheniramine (Chlor-Trimeton)
Competes with histamine or H1 receptor sites on effector cells in blood vessels and respiratory tract.
- Dosing
- Interactions
- Contraindications
- Precautions
Adult
4 mg PO q4-6h; not to exceed 24 mg/d
Pediatric
<2 years: Not established
2-6 years: 1 mg PO divided q4-6h; not to exceed 6 mg/d
6-12 years: 2 mg PO q4-6h; not to exceed 12 mg/d
>12 years: Administer as in adults
- Dosing
- Interactions
- Contraindications
- Precautions
CNS toxicity increases with coadministration of other CNS depressants, tricyclic antidepressants, MAOIs, and phenothiazines
- Dosing
- Interactions
- Contraindications
- Precautions
Documented hypersensitivity; asthma attacks; narrow-angle glaucoma; symptomatic prostate hypertrophy; bladder neck obstruction; stenosing peptic ulcer
- Dosing
- Interactions
- Contraindications
- Precautions
Pregnancy
B – Fetal risk not confirmed in studies in humans but has been shown in some studies in animals
Precautions
Drowsiness, dizziness, and dryness of mouth are the most common adverse effects; not for administration to premature or full-term neonates
Diphenhydramine (Benadryl, Belix)
For symptomatic relief of pruritus caused by endogenous release of histamine.
- Dosing
- Interactions
- Contraindications
- Precautions
Adult
25-50 mg PO tid/qid; not to exceed 400 mg/d
Pediatric
12.5-25 mg PO tid/qid or 5 mg/kg/d or 150 mg/m2/d PO divided tid/qid; not to exceed 300 mg/d
- Dosing
- Interactions
- Contraindications
- Precautions
Potentiates effect of CNS depressants; because of alcohol content, do not give syr dosage form to patient taking medications that can cause disulfiramlike reactions
- Dosing
- Interactions
- Contraindications
- Precautions
Documented hypersensitivity; MAOIs
- Dosing
- Interactions
- Contraindications
- Precautions
Pregnancy
C – Fetal risk revealed in studies in animals but not established or not studied in humans; may use if benefits outweigh risk to fetus
Precautions
Drowsiness, dizziness, and dryness of mouth are the most common adverse effects; may exacerbate angle-closure glaucoma, hyperthyroidism, peptic ulcer, and urinary tract obstruction
Topical anti-inflammatory agents
This industrial solvent has been used with mixed results.
Dimethyl sulfoxide (Rimso-50)
May help relieve symptoms. DMSO, an oxidation product of dimethyl sulfide, is an exceptional solvent possessing a number of commercial uses. Not an FDA-approved indication.
- Dosing
- Interactions
- Contraindications
- Precautions
Adult
50% solution in water applied topically
Pediatric
Not established
- Dosing
- Interactions
- Contraindications
- Precautions
None reported
- Dosing
- Interactions
- Contraindications
- Precautions
Documented hypersensitivity
- Dosing
- Interactions
- Contraindications
- Precautions
Pregnancy
C – Fetal risk revealed in studies in animals but not established or not studied in humans; may use if benefits outweigh risk to fetus
Precautions
Garliclike breath odor and taste in the mouth due to excretion of small amount of DMSO as dimethyl sulfide (usually lasts only 24-48 h)